Browse our range of reports and publications including performance and financial statement audit reports, assurance review reports, information reports and annual reports.
Department of Health’s Management of Financial Assistance under the Medical Research Future Fund

Please direct enquiries through our contact page.
Audit snapshot
Why did we do this audit?
- The Medical Research Future Fund (MRFF) is intended to improve the health and wellbeing of Australians and its administration is intended to ensure a coherent and consistent approach in the funding of medical research and medical innovation.
- The Department of Health (Health) is responsible for managing financial assistance made available from MRFF for medical research and medical innovation.
- The audit will provide assurance to the Australian Parliament and public as to how MRFF legislation and governance has guided MRFF grants for medical research and medical innovation.
Key facts
- MRFF is a $20 billion fund. Its investment returns fund medical research and medical innovation.
- In 2019 the Government committed $5.1 billion over 10 years to MRFF.
What did we find?
- Health’s management of MRFF is largely effective.
- Clear governance and coordinating structures have been established and the roles and responsibilities of Health and its implementation partners are defined. Health’s management of MRFF grants is largely compliant with legislative and policy requirements.
- Health does not have adequate performance measures for MRFF
What did we recommend?
- The Auditor-General made three recommendations to Health to identify, assess and manage risks at the theme or initiative level of the 10-year Plan, report grants in the way they are classified in the relevant grant opportunity guidelines and improve MRFF performance measures and reporting.
- Health agreed to the recommendations.
$1.8 billion
Total value of MRFF grants awarded to 30 June 2021.
670 grants
Number of MRFF grants awarded to 30 June 2021.
$1.2 billion
Total MRFF expenditure to 30 June 2021.
Summary and recommendations
Background
1. The Medical Research Future Fund (MRRF) was established in 2015 under the Medical Research Future Fund Act 2015 (MRFF Act) ‘to improve the health and wellbeing of Australians’.1
2. MRFF operates as an endowment fund with the capital preserved in perpetuity. It was established by section 11 of the MRFF Act and is managed by the Future Fund Board of Guardians.2 It was fully capitalised at $20 billion in July 2020.3 The Future Fund Board determines the maximum annual distribution amounts for each financial year from the MRFF Special Account. The Treasurer and the Minister for Finance (Finance Minister), who are the ‘responsible ministers’ under the MRFF Act, issue the investment mandate to the Future Fund Board. As required by the Minister for Health (Health Minister), funds are transferred from the MRFF Special Account to the MRFF Health Special Account, from which MRFF grants for medical research and medical innovation are made. The Department of Finance (Finance) manages the MRFF Special Account and the Department of Health (Health) manages the MRFF Health Special Account.4 As at 30 June 2021, 670 grants with a total project value of $1.8 billion had been awarded through 142 grant opportunities under MRFF.
Rationale for undertaking the audit
3. MRFF is intended to improve the health and wellbeing of Australians and ‘ensure that a coherent and consistent approach is adopted in the funding of medical research and medical innovation’.5 The Australian Government has committed to spend $5.1 billion over 10 years through 20 initiatives on the program. This audit will provide assurance to the Australian Parliament and the public about how the MRFF legislation, governance, strategies and priorities guide selection of medical research and medical innovation.
Audit objective and criteria
4. The audit objective was to assess whether Health is effectively managing financial assistance under MRFF.
5. To form a conclusion against this objective, the following high-level criteria were applied:
- Are MRFF governance arrangements effective?
- Are MRFF grants consistent with the requirements of relevant legislation and policy, including the MRFF Act?
- Has Health effectively reported on the performance of the MRFF and evaluated the effectiveness of MRFF financial assistance?
Conclusion
6. Health’s management of financial assistance under the MRFF is largely effective.
7. Health’s governance arrangements for the management of MRFF are largely effective. Clear governance roles, responsibilities and coordinating structures have been established and Health has implemented largely effective arrangements with its implementation partners to administer MRFF. Health has effectively supported consultations by the Australian Medical Research Advisory Board (AMRAB) and expert advisory panels, but it has not actively consulted with state and territory governments on the implementation of the program. The identification and documentation of the management of issues and risks should be improved.
8. Health’s management of grants of financial assistance to support medical research and medical innovation is largely consistent with the MRFF Act and the Commonwealth Grant Rules and Guidelines 2017 (CGRGs). There is no evidence of how the design of the MRFF 10-year Plan was influenced by the Australian Medical Research and Innovation Strategy (MRFF Strategy) and Australian Medical Research and Innovation Priorities (MRFF Priorities). Health has suitable arrangements in place to provide assurance that grant applications are assessed on their merit and represent value for money.
9. Health does not have adequate performance measures for MRFF and has not effectively measured and reported on the performance of MRFF financial assistance in its annual performance statements. Health published a monitoring and evaluation strategy in November 2020, with most of the activities yet to occur. It has also made a number of improvements to the operation of the program.
Support findings
Governance
10. The roles and responsibilities of the Health Minister, AMRAB, Health and its implementation partners are clearly defined. Health has agreed arrangements in place with its implementation partners and has established suitable oversight arrangements for the program. However, AMRAB has had no ministerially appointed members since March 2021 and Health has not formalised arrangements with the National Health and Medical Research Council (NHMRC) for coherent and consistent coordination of MRFF funding and NHMRC programs.
11. Stakeholder views have been considered in developing the MRFF Strategy, two-yearly MRFF Priorities and roadmaps for the research missions in the 10-year Plan. Health has not actively consulted with state and territory governments, which are key stakeholders in the delivery of MRFF and its outcomes.
12. Risks are identified and assessed at the corporate, program and grant opportunity levels in accordance with Health’s risk management policy. However, it is not always clear that issues and identified risks are being effectively managed, and risks and controls relating to the implementation of the themes and initiatives in the 10-year Plan have not been identified and managed. Health also has not consistently updated the registers of AMRAB and expert advisory panel members’ conflicts of interest and has not made them available to the public for transparency.
Grants for medical research and medical innovation
13. There is no direct relationship between the initiatives in the 10-year Plan and the MRFF Strategy and MRFF Priorities and it is not clear how the 10-year Plan was designed.
14. Health’s management of grant opportunities has been largely consistent with legislative and policy requirements, including the MRFF Act and CGRGs. However, Health has not consistently advised the Health Minister of the MRFF Priorities that the proposed grant opportunities would address and its approach to reporting the outcomes of grant opportunities reduces transparency. Health provides little information on priorities being considered for future grant opportunities.
15. Health’s processes for selecting and approving grants provide adequate assurance that grants are assessed consistently with the grant opportunity guidelines and represent value for money.
Performance management
16. The Health Minister provided reports to the Parliament on MRFF financial assistance awarded while the 2016–2018 and 2018–2020 MRFF Priorities were in force. However, Health does not have adequate performance measures for MRFF against which to report to Parliament on the performance of the program in its annual performance statements and the statements include little analysis of the program’s performance.
17. Health published a monitoring, evaluation and learning strategy for MRFF in November 2020, with most of the planned activities in the strategy yet to occur. Health has established suitable continuous improvement processes and has made a number of improvements to the operation of the program.
Recommendations
Recommendation no. 1
Paragraph 2.57
Department of Health identify, assess and manage risks at the theme or initiative level of the 10-year Plan.
Department of Health response: Agreed.
Recommendation no. 2
Paragraph 3.37
Department of Health reports grants in the same way that grant opportunities are classified in the grant opportunity guidelines and reported on GrantConnect.
Department of Health response: Agreed.
Recommendation no. 3
Paragraph 4.13
Department of Health develops adequate performance measures for the Medical Research Future Fund for inclusion in its portfolio budget statements and annual performance statements.
Department of Health response: Agreed.
Summary of Department of Health’s response
18. Health’s summary response to the report is provided below and full responses from all entities are at Appendix 1.
The Department of Health (the Department) acknowledges the findings in this report and accepts the recommendations. The Department is committed to implementing the Australian National Audit Office’s recommendations and is taking steps to address the issues identified in this audit.
It is pleasing to note the finding that the Department’s management of financial assistance under the Medical Research Future Fund (MRFF) and governance arrangements for the management of the MRFF are largely effective and consistent with legislative and policy requirements.
The Department also welcomes the finding that the Department has adequate arrangements in place to provide assurance that grant applications are assessed on their merit and represent value for money. The Department also appreciates the report’s recognition that the Department is continuously engaging and consulting with its implementation partners and key stakeholders to improve the effectiveness and operation of the MRFF.
The audit found some opportunities to improve the identification, assessment and management of risk; reporting of grants and grant opportunities; and performance measures for the MRFF. To address these findings, the Department has commenced updating its risk management processes to monitor risks at the sub-program level and is reviewing public reporting of MRFF grants. It is also developing a methodology to enable more effective reporting to the Parliament on the performance of MRFF financial assistance.
Since its inception in 2015 the MRFF has grown rapidly to become a central pillar of the Australian Government’s commitment to health and medical research. During this time, the Department has developed and implemented a range of strategies to ensure effective disbursement of MRFF funding and will use the findings of this audit to continue enhancing program arrangements.
Key messages from this audit for all Australian Government entities
Below is a summary of key messages, including instances of good practice, which have been identified in this audit and may be relevant for the operations of other Australian Government entities.
Performance and impact measurement
Governance
Risk management
1. Background
Introduction
1.1 The Medical Research Future Fund (MRRF) was established in 2015 under the Medical Research Future Fund Act 2015 (MRFF Act) ‘to improve the health and wellbeing of Australians’.6
1.2 MRFF operates as an endowment fund with the capital preserved in perpetuity. It was established by section 11 of the MRFF Act and is managed by the Future Fund Board of Guardians.7 It was fully capitalised at $20 billion in July 2020.8 Its earnings are credited to the MRFF Account. The Future Fund Board determines the maximum annual distribution amounts for each financial year from the MRFF Special Account. The Treasurer and the Minister for Finance (Finance Minister), who are the ‘responsible ministers’ under the MRFF Act, issue the investment mandate to the Future Fund Board.
1.3 Funds for the payment of grants under MRFF are transferred from the MRFF Special Account to the MRFF Health Special Account, as required by the Minister for Health (Health Minister) under section 25 of the MRFF Act. The MRFF Special Account is managed by the Department of Finance (Finance).9 The Department of Health (Health) is responsible for managing the MRFF Health Special Account and financial assistance under MRFF, including dispersing funds for medical research and medical innovation.10
1.4 The Australian Medical Research Advisory Board (AMRAB), established under section 32B of the MRFF Act, is required under section 32D to determine an Australian Medical Research and Innovation Strategy (MRFF Strategy) every five years to help ensure that a coherent and consistent approach is adopted in providing financial assistance under the MRFFF Act for medical research and medical innovation. Section 32E also requires AMRAB to develop Australian Medical Research and Innovation Priorities (MRFF Priorities) that are consistent with the MRFF Strategy every two years.11
1.5 The six strategic platforms identified in the 2016–2021 MRFF Strategy and the MRFF Priorities are shown at Table 1.1.12
Table 1.1: 2020–22 MRFF Priorities by strategic platform
|
2016–2020 MRFF Strategy strategic platforms |
2020–22 MRFF Priorities |
|
Strategic and international horizons |
One Health – Antimicrobial resistance |
|
Global health and security |
|
|
Aboriginal and Torres Strait Islander health |
|
|
Ageing and aged care |
|
|
Data and infrastructure |
Digital health intelligence |
|
Health services and systems |
Comparative effectiveness research |
|
Primary care research |
|
|
Capacity and collaboration |
Clinical researcher capacity |
|
Consumer-driven research |
|
|
Trials and translation |
Drug repurposing |
|
Public health interventions |
|
|
Commercialisation |
Translational research infrastructure |
Source: Australian Medical Research and Innovation Priorities 2020–2022.
1.6 In the Budget the Australian Government announced a 10-year Plan for MRFF investments. The 10-year Plan consists of planned expenditure of $5.1 billion from 2018–19 to 2027–28 on medical research and medical innovation research through 20 initiatives, which are grouped into the four themes shown in Table 1.2.
Table 1.2: Funding themes in the 10-year Plan
|
Theme |
Description |
|
Patients |
Funding innovative treatments, supporting clinical trials, and delivering more advanced health care and medical technology to improve the health of all Australians. |
|
Researchers |
Supporting our researchers to make breakthrough discoveries, develop their skills and progress their careers in Australia. |
|
Research missions |
Helping researchers think big to tackle significant health challenges through investment, leadership and collaboration. |
|
Research translation |
Moving research ideas from the lab to the clinic, so that medical discoveries become part of clinical practice for GPs, specialists and hospitals. |
Source: Department of Health, Medical Research Future Fund 10-year Plan.
1.7 Health is responsible for the overall administration of MRRF grants, with support from its implementation partners — the Department of Industry, Science and Energy Resources (Industry), the National Health and Medical Research Council (NHRMC) and Cancer Australia. With some exceptions, approved grant opportunities are managed for Health by either NHMRC or Industry’s Business Grants Hub (BGH). They act as the grants hubs for the program.13
1.8 As presented in its Portfolio Budget Statements, Health supports the government to create a better health system and improve the wellbeing of all Australians through six outcomes and 27 programs. The MRFF program, under Program 1.1 Health Research, Coordination and Access, is a key contributor to Outcome 1: Health Policy, Access and Support.
Grants provided through the Medical Research Future Fund
1.9 As at 30 June 2021, 670 grants with a total project value of $1.8 billion had been awarded through 142 grant opportunities under MRFF. The number and value of grants awarded by 10-year funding theme in the 10-year Plan as at 30 June 2021 are shown in The number and value of grants awarded by 10-year Plan initiative as at 30 June 2021 are shown in Appendix 3 and the actual and budgeted amounts against the 10-year Plan are shown at Appendix 4.
Table 1.3: Number and value of MRFF grants by 10-year Plan theme as at 30 June 2021
|
|
All grantsa |
||
|
Theme |
Value |
Number |
|
|
Patients |
$564,846,991 |
220 |
|
|
Researchers |
$238,615,326 |
135 |
|
|
Research missions |
$426,595,385 |
179 |
|
|
Research translation |
$549,780,009 |
136 |
|
|
Total |
$1,779,837,711 |
670 |
|
Note a: Includes all MRFF grants awarded and announced since the program’s inception in 2015 and the total value of grant agreements across all financial years, including forward years. Some contracts for awarded grants had not been executed as at 30 June 2021. Also includes 53 grants with a value of $68.63 million funded as part of the Government’s Coronavirus Research Response.
Source: Department of Health.
1.10 Payments made for approved grants from 2016–17 to 2020–21 are shown in Table 1.4.
Table 1.4: Payments made for MRFF grants, 2016–17 to 2020–21a
|
Theme |
2016–17 $m |
2017–18 $m |
2018–19 $m |
2019–20 $m |
2020–21 $m |
Total $m |
|
Patients |
7.960 |
7.283 |
112.541 |
125.463 |
150.860 |
404.107 |
|
Researchers |
– |
1.815 |
4.919 |
47.881 |
84.005 |
138.620 |
|
Research missions |
– |
1.002 |
44.732 |
120.675 |
185.610 |
352.019 |
|
Research translation |
10.000 |
20.000 |
44.174 |
81.505 |
177.460 |
333.139 |
|
Total |
17.960 |
30.100 |
206.366 |
375.524 |
597.935 |
1,227.885 |
Note a: Amounts reflect payments made in each financial year, not the total value of grant agreements finalised in those years.
Source: Department of Health.
Rationale for undertaking the audit
1.11 MRFF is intended to improve the health and wellbeing of Australians and its ‘administration will ensure that a coherent and consistent approach is adopted in the funding of medical research and medical innovation’.14 The Australian Government has committed to spend $5.1 billion over 10 years through 20 initiatives on the program. This audit will provide assurance to the Australian Parliament and the public about how the MRFF legislation, governance, strategies and priorities guide selection of medical research and medical innovation.
Audit approach
Audit objective, criteria and scope
1.12 The audit objective was to assess whether Health is effectively managing financial assistance under MRFF.
1.13 To form a conclusion against this objective, the following high-level criteria were applied:
- Are MRFF governance arrangements effective?
- Are MRFF grants consistent with the requirements of relevant legislation and policy, including the MRFF Act?
- Has Health effectively reported on the performance of MRFF and evaluated the effectiveness of MRFF financial assistance?
Audit methodology
1.14 The audit methodology involved:
- examining Health’s MRFF records;
- assessing compliance with MRFF legislation;
- mapping changes to the MRFF Priorities over time and their alignment with the MRFF Strategy and grant opportunities;
- reviewing a sample of 69 MRFF grants from a population of 482 grants awarded under 46 grant opportunities as at 2 December 2020;
- consulting with MRFF partner entities (Industry, NHRMC and Cancer Australia) and other stakeholders (Finance and two AMRAB members);
- interviewing relevant Health staff; and
- considering 33 submissions to the ANAO, which included submissions from industry bodies, universities, research institutes, advocacy groups and individual researchers.
1.15 The audit was conducted in accordance with ANAO Auditing Standards at a cost to the ANAO of approximately $475,000.
1.16 Team members for this audit were John McWilliam, Fraser McEachan, Clarina Harding, Dr Cristiana Linthwaite-Gibbins and Deborah Jackson.
2. Governance
Areas examined
This chapter examines the effectiveness of the Department of Health's (Health's) governance arrangements for administering Medical Research Future Fund (MRFF) grants.
Conclusion
Health's governance arrangements for the management of MRFF are largely effective. Clear governance roles, responsibilities and coordinating structures have been established and Health has implemented largely effective arrangements with its implementation partners to administer MRFF. Health has effectively supported consultations by the Australian Medical Research Advisory Board (AMRAB) and expert advisory panels, but it has not actively consulted with state and territory governments on the implementation of the program. The identification and documentation of the management of issues and risks should be improved.
Areas for improvement
The ANAO has made one recommendation aimed at ensuring that risks at the initiative or theme level of the 10-year Plan are identified and managed.
The ANAO has also suggested that Health actively consult with state and territory governments in relation to MRFF financial support.
2.1 Governance involves the systems and processes in place that shape, enable and oversee the management of an organisation. Under the Public Governance, Performance and Accountability Act 2013 (PGPA Act), an accountable authority of a Commonwealth entity must govern the entity in a way that promotes: the proper use and management of public resources for which the authority is responsible; achievement of the purposes of the entity; and financial sustainability of the entity.15
2.2 To assess whether Health's governance arrangements were effective, the ANAO examined:
- the clarity of roles and responsibilities designed to provide oversight of MRFF funding activities;
- Health's arrangements to consider and address stakeholder views; and
- Health's arrangements to identify, assess and manage risk.
Are the roles and responsibilities for the Medical Research Future Fund clearly defined?
The roles and responsibilities of the Minister for Health, AMRAB, Health and its implementation partners are clearly defined. Health has agreed arrangements in place with its implementation partners and has established suitable oversight arrangements for the program. However, AMRAB has had no ministerially appointed members since March 2021 and Health has not formalised arrangements with NHMRC for coherent and consistent coordination of MRFF funding and NHMRC programs.
2.3 Clearly defined roles and responsibilities in cross-functional entity programs are important for understanding accountability and who should be consulted or informed of program developments and outcomes.
2.4 The ANAO examined the roles and responsibilities of Ministers, the AMRAB, Health and its key implementing partners.
Ministerial roles and responsibilities
2.5 The Medical Research Future Fund Act 2019 (MRFF Act) sets out the roles and responsibilities of the:
- Minister for Finance (Finance Minister) and the Treasurer, who are both responsible for oversight of the Medical Research Future Fund; and
- the Minister for Health (Health Minister), who is responsible for administration of MRFF financial assistance to support medical research and innovation.
2.6 The Health Minister, when requiring the Finance Minister to debit the MRFF Special Account to channel funds to a grant recipient, must take into account the MRFF Priorities that are in force. The Health Minister also has regard to the 10-year Plan, which details the government's financial commitments to support MRFF.
Australian Medical Research Advisory Board
Appointments to the Australian Medical Research Advisory Board
2.7 Under sections 32F and 32G of the MRFF Act, the membership of AMRAB consists of the chief executive officer of NHMRC and up to seven other members appointed as part-time members by the Health Minister. AMRAB's terms of reference are consistent with the MRFF Act.
2.8 The maximum term for the ministerially appointed AMRAB members is five years. The membership of ministerially appointed members expired on 28 February 2021 (one member resigned in 2020). Health advised the Health Minister in November 2020 that the terms of the AMRAB members were due to expire in February 2021 and obtained his agreement to revised terms of reference and a process to fill the positions by March 2021. New members have not yet been appointed.
2.9 A person is not eligible for appointment to AMRAB under subsection 32G(2) unless the Health Minister is satisfied that, if the person were appointed, the Board would collectively possess an appropriate balance of experience or knowledge in eight nominated fields. Table 2.1 summarises the ANAO's assessment of the collective experience or knowledge of the previous Board members in those fields.
Table 2.1: Collective experience or knowledge of AMRAB members, 2016–2021
|
Field of experience or knowledge |
Overall composition |
|
Medical Research |
|
|
Policy relating to health systems |
|
|
Management or delivery of health services |
|
|
Medical innovation |
|
|
Financing and investment |
|
|
Commercialisation |
|
|
Philanthropy |
|
|
Consumer issues relating to health |
|
Key:
![]() Very high level representation
Very high level representation
![]() High representation
High representation
![]() Medium representation
Medium representation
![]() Low representation
Low representation
Source: ANAO analysis of biographies provided by Health.
Role and responsibilities of the Australian Medical Research Advisory Board
2.10 Under section 32D of the MRFF Act, AMRAB's role is to determine an Australian Medical Research and Innovation Strategy (MRFF Strategy) every five years for ensuring that a coherent and consistent approach is adopted in providing financial assistance under the MRFF Act for medical research and medical innovation. AMRAB is also required to develop Australian Medical Research and Innovation Priorities (MRFF Priorities) every two years, consistent with the MRFF Strategy.
2.11 In determining MRFF Priorities, section 32F of the MRFF Act requires AMRAB to take into account:
- the burden of disease on the Australian community;
- how to deliver practical benefits from medical research and medical innovation to as many Australians as possible;
- how to ensure that financial assistance provided under this Act provides the greatest value for all Australians;
- how to ensure that financial assistance provided under this Act complements and enhances other financial assistance provided for medical research and medical innovation; and
- any other relevant matter.
2.12 AMRAB released the first MRFF Strategy for 2016–2021 on 9 November 2016 and has also published Priorities for 2016–2018, 2018–2020 and 2020–22. Because AMRAB membership (other than the chief executive officer of NHMRC) has lapsed, work has not yet commenced on the MRFF Strategy for 2021–2026.
2.13 Under paragraph 32C(b) of the MRFF Act, a function of AMRAB is to advise the Health Minister about matters other than the MRFF Strategy and MRFF Priorities that the Minister refers to it. The Minister has not sought AMRAB's advice on other matters. The changes to AMRAB's terms of reference, which the Health Minister approved in November 2020, include matters on which AMRAB will be asked to provide advice to the Minister. The terms of reference for AMRAB are not publicly available.16
2.14 AMRAB met fourteen times from 4 May 2016 to 20 October 2020, meeting twice each financial year, with the exception of 2017–18, the first full year of AMRAB's operations, when it met four times. Communiqués on two of AMRAB's meetings (26 July 2019 and 12 March 2020) are available on Health's website.17 Draft communiqués were prepared for other AMRAB meetings, but were not finalised and published.
Health's role and responsibilities
2.15 Health's role is to support the Health Minister and AMRAB in the implementation of the MRFF Act and to effectively administer MRFF grants. It:
- develops and, where necessary, obtains the Health Minister's approval to supporting policies, such as the MRFF funding principles, and other procedures;
- develops and obtains the Health Minister's approval of all new MRFF grant opportunities;
- manages all new grant opportunities, including through its implementation partners;
- exercises the Health Minister's delegation to approve grants based on the assessments of grant assessment committees;
- monitors the progress of projects by grant recipients, including through its implementation partners, and approves progress reports;
- provides secretariat support to AMBAB and other consultative or advisory bodies (in particular, expert advisory panels or committees appointed by the Minister to advise on the Mission initiatives in the 10-year Plan);
- consults stakeholders, as needed, on the implementation of the program; and
- reports on the performance of the program.
2.16 Within Health, the Health and Medical Research Office (HMRO) was established in the Budget, with a departmental budget of $5 million a year from 2019–20 to 2022–23, to manage financial assistance under MRFF.18
2.17 Table 2.2 lists the key instruments that Health is required to follow in the administration of MRFF.
Table 2.2: MRFF key governance instruments
|
Instrument |
Summary description |
|
Administrative Arrangements Order – 18/03/2021 |
Health's responsibilities include medical, health and ageing research. |
|
MRFF Act, sections 15A, 20, 21, 22, 25, 26, 27, 29 and 30 |
All disbursements must occur in accordance with the MRFF Act. The Health Minister must take the MRFF Priorities into account when making decisions of financial assistance to support medical research and medical innovation. |
|
Health Minister instrument of delegation for MRFF Act, section15A |
Requires the Finance Minister to debit a specified amount from the Medical Research Future Fund Special Account. Approved delegates are:
|
|
Health Minister's instrument of delegation for MRFF Act, sections 26 and 27 |
As soon as practicable the Minister must ensure the MRFF Health Special Account is debited for the purpose of making one or more grants, and entering the grant agreement on behalf of the Commonwealth. Approved delegates are:
|
|
MRFF Funding Principles |
Guide the allocation of MRFF research funding Developed by AMRAB, endorsed by the Health Minister. |
|
Department of Health Corporate Plana |
Program 1.1. Health System Policy, Design and Innovation of Outcome 1: Health System Policy, Design and Innovation. |
Note a: The outcomes and programs shown are for 2020–21. The 2021–22 PBS includes the new outcomes and programs (see paragraph 1.8).
Source: ANAO analysis of governance instruments.
Program Assurance Group
2.18 Following an internal audit of Health's implementation of MRFF (see paragraph 4.23), Health established a Program Assurance Group (PAG) in October 2019 to provide internal oversight of its management of MRFF. PAG's chair is the First Assistant Secretary, Health Economics and Research, and the deputy chair is the head of HMRO. There are nine other members including an executive from Cancer Australia.
2.19 PAG's terms of reference state that its role 'is to provide assurance to MRFF implementation and to assist with the continuous improvement of assurance mechanisms, risk management practices and program assurance more broadly as a result of lessons learnt and to increase consistency across MRFF initiatives'. It:
- advises on the acceptable risk profile and risk thresholds for the MRFF and its initiatives;
- considers strategic directional issues for MRFF initiatives;
- reviews risk, cost, quality and timelines for each of the MRFF themes and initiatives; and
- advises on program assurance issues to improve the alignment of activities and consistency of processes that are essential to optimal program performance and ensuring the delivery of the MRFF program outputs and outcomes.
2.20 To assess whether PAG has effectively undertaken the role set out in its terms of reference, the ANAO reviewed the agenda, meeting papers and outcomes of meetings held between October 2019 and 4 December 2020. The ANAO found that PAG met monthly and considered strategic and operational papers and agenda items that could directly impact the success of the MRFF program. Meeting outcomes were documented in an action register that was reviewed at the start of each meeting and updated at the end of each meeting. The outcomes should be reported to the relevant Health deputy secretary at least quarterly. Health advised that PAG has provided two reports to the associate secretary.
2.21 Overall, the ANAO found that PAG provides a suitable forum for coordination of MRFF responsibilities within Health and oversight of the program.
Health's arrangements with its implementation partners
2.22 In their roles as grants hubs for the MRFF, NHMRC and the Department of Industry, Science, Energy and Resource's (Industry) Business Grants Hub (BGH):
- review grant opportunity guidelines drafted by Health;
- prepare risk assessments for the grant opportunities and, where required, arrange clearance of these with the Department of Finance (Finance) and the Department of the Prime Minister and Cabinet;
- arrange for grant opportunities to be issued through GrantConnect;
- manage assessments of grants in response to grant opportunities (with grant assessment committees being established to review the grant applications);
- arrange for grant agreements with successful applicants to be prepared and approved; and
- provide day-to-day oversight of the performance of grant projects, including advising Health on acceptance of progress reports.
2.23 Cancer Australia also provided grants hub services for some early grant opportunities, but ceased doing so in 2018. Its role is to support Health by advising on priority areas of cancer research and related grant opportunities, and by advising on the adequacy of progress reports provided by grant recipients and any follow-up action that should be considered.
2.24 Health has memoranda of understanding (MoUs) with NHMRC and Industry on the provision of grants hub services and with Cancer Australia for its support on the Brain Cancer Mission.19 Each of these MoUs articulate:
- the objectives of the arrangement, including desired outcomes and timeframes;
- the roles and responsibilities of the parties;
- the details of the activities, including specifications of services or projects to be undertaken;
- resources and timeframes to be applied by parties;
- the approach to identifying and sharing the risks and opportunities involved;
- agreed modes of review and evaluation; and
- agreed dispute resolution arrangements.
2.25 Health has fortnightly operational meetings and quarterly strategic meetings with NHMRC and BGH. It also has regular meetings with Cancer Australia on the brain cancer mission. These meetings identify and consider issues and are the primary communication mechanisms between Health and its implementation partners. Each of the three implementation partners considered these arrangements work well.
2.26 The roles and responsibilities of Health, NHMRC and BGH have evolved over the first five years of the program. Health now exercises greater responsibility for the development of grant opportunities and approval of grantees' progress reports. This reflects, among other things, increased resourcing for MRFF administration following the establishment of the HMRO in 2019 and Health's advice to the ANAO that it is seeking to take a more active role in monitoring the performance of grant projects. In this context Health has implemented a common reporting template for the MRFF program to provide greater consistency of progress reporting by grant recipients across both grants hubs.
2.27 The MRFF Act allows the Health Minister to delegate functions to Health and NHMRC but not to BGH. This has resulted in differences in MRFF administrative processes. Grant agreements are executed by NHMRC through an instrument of delegation to enter agreements on behalf of the Commonwealth, after Health has approved the grants. Agreements managed by BGH are approved and signed by Health.
2.28 The MoU with NHMRC was signed in 2017 and revised in May 2021. The revised MoU clarified the respective roles, responsibilities and management of shared risks. The MoUs with Industry and Cancer Australia were signed in 2018. The relevant schedule to the MoU with Industry is updated for each grant opportunity. The MoU with Cancer Australia requires updating to reflect the fact that it is no longer providing grants hub services.
2.29 Under paragraph 32D(3)(a) of the MRFF Act, in preparing the five-year MRFF Strategy AMRAB must take into account the 'national strategy for medical research and public health research prepared for the purposes of paragraph 16(2)(c) of the National Health and Medical Research Council Act 1992'. AMRAB included a chapter on 'alignment and complementarity', with a section on NHMRC, in the MRFF Strategy. In the Strategy AMRAB stated that:
The relationship between the MRFF and NHMRC will evolve through collaboration, facilitated by the welcome inclusion of the NHMRC Chief Executive Officer on the Advisory Board. …
Both the NHMRC and the Advisory Board are committed to working together to ensure complementarity of funding is maintained through collaboration, governance, and shared administration where appropriate.20
2.30 NHMRC describes its role as:
the Australian Government's primary health and medical research funding agency. With NHMRC support, Australia undertakes outstanding health and medical research which has contributed to significant improvements in individual and population health.21
2.31 Health's MoU with NHMRC is silent on arrangements for coordination of MRFF support with grants administered by NHMRC. Both NHMRC and Health (through MRFF) provide grants for health and medical research to recipients from the same organisations. There is some overlap between the two programs, particularly in relation to the awarding of research fellowships. MRFF fellowships were awarded in 2019 and 2020 using NHMRC peer review outcomes for NHMRC investigator grants.22
2.32 To strengthen links between the two programs and manage potential overlaps between them, Health could usefully include mechanisms for coordination of activities in future updates of the MoU with NHMRC. Such coordination might, for example, include an analysis of the outcomes of research being conducted by NHMRC grant recipients and any implications for future MRFF research priorities.
2.33 The former chair of AMRAB advised the ANAO that he considered that there is a need for MRFF to be supported by an organisation with a similar structure and capacity to that of NHMRC and expert advisory committees, and a business team that would manage potential overlaps between MRFF, NHMRC and Industry.
Monitoring grant recipient progress
2.34 As part of their MRFF grant agreements, grant recipients are required to give Health progress reports during the course of projects and final reports on completion of projects.23 Progress reports are designed to ensure that satisfactory progress is being made before further grant instalment payments are made. Health and its implementation partners review grantee progress reports to assess whether satisfactory progress has been made and identify issues that may need to be followed up with the grant recipient.24 In the 69 grants sampled by the ANAO, two projects had been completed and final reports had been submitted and two fellowships had been completed.
Are relevant stakeholder views considered and addressed where appropriate?
Stakeholder views have been considered in developing the MRFF Strategy, two-yearly MRFF Priorities and roadmaps for the research missions in the 10-year Plan. Health has not actively consulted with state and territory governments, which are key stakeholders in the delivery of MRFF and its outcomes.
2.35 Consideration of stakeholder views is important for determining research priorities and assuring that MRFF grant opportunities and grants are managed effectively. Health supports AMRAB's consultation on the MRFF Strategy and MRFF Priorities and the expert advisory panels in their consultation on the roadmaps and implementation plans for the 10-year Plan missions. Having an effective communications strategy can help to facilitate a successful consultation process.
Communications strategy and plan
2.36 AMRAB endorsed a MRFF website and communication strategy at its 10th meeting on 26 February 2019, which includes a budget and timeframes for implementing the planned activities. The strategy sets out a two-tiered approach to communication planning:
- planned communication activities that would run at various times of the year to inform and promote specific initiatives, funding opportunities and events; and
- ongoing news activities that underpin the planned communication with ongoing delivery of key messages to target audiences.
2.37 This strategy resulted in redesigned MRFF web pages and 20 'snapshots' (factsheets) on each of the initiatives under the 10-year Plan.
2.38 In March 2020, Health adopted a MRFF communication plan to:
- provide consistent, timely and accurate information about MRFF grant opportunities and outcomes;
- promote the achievements of the MRFF, particularly research outcomes; and
- increase awareness and understanding of the MRFF.
2.39 Health's website is one of the key channels for MRFF messaging. The website includes information about the 10-year Plan, MRFF Strategy and MRFF Priorities, the 20 initiatives set out in the 10-year Plan, as well as a calendar of MRFF grant opportunities and feature MRFF grant stories. Stakeholders can subscribe to a monthly MRFF newsletter via the website. However, many of the MRFF initiative snapshots on the website were last updated in November 2019 and the AMRAB page has not been updated since November 2020.
Health's support for the Australian Medical Research Advisory Board and expert advisory panel consultation
2.40 Under section 32EA of the MRFF Act, AMRAB is required, when determining a MRFF Strategy or MRFF Priorities, to provide a process for consulting:
- organisations with expertise in medical research or medical innovation;
- organisations that represent consumers who benefit from medical research or medical innovation; and
- any other person or organisation.
2.41 Health provided support to AMRAB for its consultation on the MRFF Strategy (2016-2021) and each of the three MRFF Priorities (2016–2018, 2018–2020 and. Health's support included preparation of a consultation strategy, drafting supporting papers for AMRAB to use as a basis for community feedback and organisation of consultative meetings and webinars. Support did not include a systematic or evidence-based assessment of healthcare needs or unmet needs and risks.
2.42 The eight expert advisory panels established by the Health Minister 'to guide' the MRFF 10-year Plan's research missions have largely consistent terms of reference. Their terms of reference state that:
- panel members are to consult and engage with other researchers, industry and consumer/patient groups; and
- participate in media and public activities to build awareness of, and facilitate interaction with, the mission and with other MRFF funded research.
Health provided support to the expert advisory panels to undertake their activities, including on the road maps and implementation plans for the missions.
2.43 Health has a consultation hub (an internet portal) for stakeholders to find, share and participate in consultation, including by providing written submissions. The hub standardises how and where stakeholders can understand and contribute to consultation. It is has been used by Health for 12 MRFF consultation processes to support AMRAB's consultation on the MRFF Strategy, the three MRFF Priorities and six mission specific roadmaps and implementation plans. Consultation has been extensive and has included reviews of the roadmaps and implementation strategies by an international panels of experts.25
Consultation with state and territory governments
2.44 The state and territory governments are key stakeholders identified in the MRFF Act as potential recipients of MRFF funds channelled through the COAG Reform Fund. State and territory governments are the major providers of healthcare in Australia, consumers of medical research and medical innovation, and are potential co-funders of MRFF grants. They are identified as key stakeholders in the MRFF communications plan. Health's corporate plans have also identified risks relating to the state and territory governments in delivering medical research funding (see Table 2.3 below).
2.45 In the MRFF communication plan, state and territory governments appear alongside 'other Commonwealth agencies' and 'health portfolio agencies'. The plan outlines targeted methods of communication for consultation with state and territory ministries and departments, including face-to-face meetings, teleconferences and committee meetings of the Health Council.26 Health has not used these methods of communication for its consultation with the state and territory governments on MRFF. Some state and territory government entities did, however, provide feedback as part of AMRAB's consultation on the MRFF Strategy and MRFF Priorities. For AMRAB's consultation on the MRFF Priorities for 2020–2022, one state and one territory made written submissions and another state raised a question during a webinar. The webinar question asked about the avoidance of duplication and collaboration and the need for a platform to find research being undertaken around the country.
2.46 As key stakeholders, Health should actively consult with state and territory governments in the implementation of MRFF.
Are risks identified, assessed and managed?
Risks are identified and assessed at the corporate, program and grant opportunity levels in accordance with Health's risk management policy. However, it is not always clear that issues and identified risks are being effectively managed, and risks and controls relating to the implementation of the themes and initiatives in the 10-year Plan have not been identified and managed. Health also has not consistently updated the registers of AMRAB and expert advisory panel members' conflicts of interest and has not made them available to the public for transparency.
2.47 The Commonwealth Risk Management Policy requires accountable authorities to endorse an entity's risk management policy and framework. Health's risk framework sets out Health's approach to the management of risk, key accountabilities and responsibilities for risk management, risk appetite and risk tolerance. It includes:
- a risk management policy, a glossary of terms and definitions, and accountable authority instructions;
- enterprise risk appetite — outlines where Health is willing to engage with higher levels of risk for a greater benefit and to achieve strategic objectives;
- enterprise level risks — those risk that potentially have the most significant impact on Health's strategic priorities and operations; and
- risk registers — live documents that are updated throughout the life of the program or project using a risk register workbook template.
2.48 To examine how effectively Health is identifying, assessing and managing MRFF risks, the ANAO examined Health's management of:
- program risks;
- grant opportunity risks;
- risks it shares with implementing partners; and
- conflicts of interest.
Program risks
2.49 MRFF contributes to Health's Outcome 1. Outcome 1 risks and management strategies identified in Health's corporate plans for 2019–20 and 2020–21 are shown at Table 2.3.
Table 2.3: Outcome 1 risks, 2019–20 and 2020–21
|
Key risks |
Management strategies |
|
2019–20 Corporate Plan |
|
|
The implementation of the ten-year Medical Research Future Fund investment plan and other MRFF initiatives is delayed. The states and territories are delayed in implementing important clinical trial system redesign strategies. |
We will apply rigorous project management processes and establish governance arrangements. We will proactively engage with internal and external stakeholders to ensure collaboration and management of expectations. |
|
2020–21 Corporate Plan |
|
|
Arrangements do not support the states and territories to deliver improved safety and quality, health innovation and sustainable hospital funding, including medical research funding. Investments in health and medical research from the MRFF are not made in accordance with legislative requirements, the MRFF 10 year Investment Plan, the Australian Medical Research and Innovation Priorities 2020–22 and the Australian Medical Research and Innovation Strategy 2021–26. |
Funding provisions under the new National Health Reform Agreement for the period to 2024–25 will increase over the coming years. This funding will continue to focus on reducing unnecessary hospitalisations and improving patient safety and service quality. Effective financial management and program assurance activities are implemented and reviewed regularly. Independent expert advice will support MRFF investment policy development. |
Source: Health Corporate Plan 2019–20 and 2020–21.
2.50 Health's Economic and Research Division, which has responsibility for MRFF, has identified the following key MRFF risk in its 2020–21 Business and Risk Planning:
Rapid growth of Medical Research Future Fund could impact effective deployment of funds to maximise outcomes and/or financial management and Health's Departmental funding and ASL positions.
2.51 In September 2020 PAG endorsed a MRFF risk register, which identifies MRFF program risks that need to be managed and mitigation strategies or controls for the risks. There are three risks identified in the register:
- MRFF investment — MRFF research investments do not achieve priorities established by Government;
- MRFF administration — MRFF funds are not deployed in accordance with legislative and policy requirements; and
- harm from a MRFF funded project — a researcher or research participant is harmed while participating in a MRFF funded research project.
2.52 Examination of the PAG meeting papers indicates that the register is included in the papers for each meeting. The format of the register is consistent with the requirements of Health's Risk Management Policy. However, it is not always clear that issues and identified risks are being effectively managed as the risk register does not include:
- sections to track and control version updates, document who is responsible for the register or record when it was endorsed by PAG or other committees; and
- an issues register 'to record all unplanned situations that are happening now that required management attention', as required by the instructions for using the register.
2.53 To ensure that it is actively managing risks, the ANAO suggests that Health updates the risk register to include the elements outlined above.
2.54 The risk appetite for each of the risks in the risk register has been assessed as medium and has been classified under a 'Delivery' theme.27 The PAG minutes and risk register do not indicate what consideration was given to classifying risks. A change of theme may alter the risk appetite and the level of control required.
2.55 The ANAO's assessment of whether the controls for the three risks in the risk register are in place is shown in Table 2.4. It is not evident from the PAG meeting documents that PAG monitors each control.
Table 2.4: Assessment of MRFF risk controls in the MRFF risk register
|
Risk |
Current mitigation strategy (control) |
Assessment |
|
MRFF Investment |
Independent expert advice to support MRFF investments (e.g. Australian Medical Research Advisory Board, Mission expert advisory committees, and ad hoc working groups). |
◆ See paragraphs 2.42 to 2.45 and paragraphs 3.11 to 3.15 |
|
Grant guidelines and policies clearly articulate expectations of grant opportunities and funded projects and are aligned with MRFF priorities (e.g. the MRFF 10-year plan). |
▲ See paragraphs 3.8 to 3.15 |
|
|
Expert review of applications against stated objectives in guidelines. |
◆ See paragraphs 3.47 to 3.55 |
|
|
Consultation across government (e.g. Research Funders Forum) and with the sector (e.g. consumers and the public) to understand key issues and support policy development. |
▲ See paragraphs 2.37 to 2.48. Health also initiated the establishment of the Research Funds Forum to enable experiences in grant management to be shared across entities. |
|
|
MRFF administration |
HMRO is developing and implementing consistent policies and procedures for administration of the MRFF. |
◆ See paragraphs 4.23 to 4.25. |
|
HMRO reports regularly on program and financial management of the MRFF (e.g. to Minister, Executive, internal and external audit committees, Program Assurance Group, Program Assurance Committee). |
◆ See paragraphs 2.19 to 2.22. |
|
|
MRFF administration (continued) |
HMRO communicates regularly with grant hubs. |
◆ See paragraphs 2.23 to 2.33 |
|
HMRO is recruiting staff and investing in training opportunities to continue improving staff capability. |
▲ There is no specific training on grants management and this issue is not being managed by PAG. |
|
|
Harm from a MRFF funded project |
Grant guidelines and funding agreements articulate safety, ethics, risk and regulatory requirements of grant opportunities and funded projects. |
◆ The review of a sample of grants indicated that the grant opportunity guidelines adequately cover these matters. |
|
Progress reports from grantees are submitted regularly and reviewed by HMRO. |
◆ See paragraphs 2.27 and 2.34. Progress reports were also reviewed as part of the audit sample. |
|
Key: ◆ Control in place
▲ Control partially in place
Source: ANAO analysis of Health documents.
2.56 PAG does not systematically identify, assess and manage risks at the initiative or theme level of the 10-year Plan. The initiatives have different risks. For example, expert advisory panels are exclusive to the missions, which would have different financial and conflict of interest risks from other initiatives. The ANAO recommends that Health, when preparing updates for PAG, includes a risk assessment for each theme or initiative to help oversee and manage these risks.
Recommendation no.1
2.57 Department of Health identify, assess and manage risks at the theme or initiative level of the 10-year Plan.
Department of Health response: Agreed.
2.58 The Department is updating its risk management processes to ensure appropriate identification, assessment and management of risks at the sub-program level. This will complement current processes already in place to monitor risks at the program level.
Grant opportunity risks
2.59 Risk assessments are undertaken for grant opportunities other than one-off or ad hoc grants. The grants hubs consider the risks associated with each grant opportunity and determine the risk rating in consultation with Health and Finance. Where the grant opportunity risk is assessed as 'medium' or above, the Finance Minister approves the grant opportunity guidelines. The ANAO reviewed a sample of seven competitive grant opportunity risk assessments between 2017 and 2020. Each consistently identified risk, the source of the risk, pre mitigation risk rating, actions to mitigate risk and a post mitigation risk rating. The context and preamble of each risk assessment was matched to the grant opportunity. Given the differing nature of many grant opportunities, the risks are likely to vary. However, the same risk assessment template and risk description were used for each grant opportunity.
Shared risks
2.60 The Commonwealth Risk Management Policy states that each entity must implement arrangements to understand and contribute to the management of shared risks.28 Health defines shared risk as a risk with no single owner, where more than one entity is exposed to or can significantly influence the risk.
2.61 Health shares some MRFF responsibilities with its implementation partners. It has highlighted to NHMRC the complementary nature of MRFF with NHMRC's Medical Research Endowment Account and noted the overlapping responsibilities and shared interests of AMRAB and the NHMRC Council (which provides advice to NHMRC's chief executive officer in relation to the performance of their functions). In 2018 NHMRC Council members noted ongoing health and medical research community concerns about the transparency of the disbursement of MRFF funds, suggesting a need for better coordination between Health and NHMRC.
2.62 A recent update to the MoU with NHMRC has addressed shared risks. The MoU with BGH was updated in 2019 with an assurance framework schedule that identifies key risks and mitigation strategies.
2.63 Health has developed escalation protocols and procedures for identification and communication of high-risk issues. The escalation channels align to the grants operating models for NHMRC and BGH. Examination of the minutes of operational meetings with NHMRC and BGH indicated that there was no discussion of shared risks or how controls affect their respective enterprise and program risk profiles. However, in mid-2021 Health, NHMRC and BGH agreed to discuss shared risks and controls at their quarterly strategic meetings and follow up actions at their fortnightly operational meetings.
Conflicts of interest
2.64 Conflicts of interest can result in a failure to act in the best interest of the entity or program and poor governance. Section 29 of the PGPA Act places a duty on all officials to disclose material personal interests relating to the affairs of the entity. It is fundamental to good governance that material personal interests are raised and dealt with effectively. Failure to do so can undermine confidence and trust in the entity and potentially the Commonwealth more broadly. Appropriate management of conflicts of interest requires a sound framework along with an active management approach (including adequate oversight of the actions of individual employees).
Management of conflicts of interest by Health staff
2.65 Health requires employees to disclose any material personal interest that relates to the affairs of the department. The overriding principle for a declaration of a material personal interest is, if in doubt, declare the interest in accordance with legislative requirements and instructions of the Secretary. Where a declaration of a personal interest is declared, the supervisors of officials making a declaration must develop and maintain a written risk management plan for that risk, which clearly defines the responsibility for managing the risk and outlines the actions to be taken to minimise potential conflicts of interest.
2.66 In March 2021 Health established a declaration of interest policy and register for Health officials involved in the administration of the MRFF. As at 20 May 2021, two members of staff were assessed as having a conflict of interest and mitigation strategies were implemented for those staff.
Management of conflicts of interest by advisors
2.67 MRFF uses a large number of advisors, either through AMRAB or the expert advisory panels, who as researchers or representatives of research organisations may submit applications for MRFF funding.
2.68 Health's Medical Research Future Fund Declaration of Interest Policy Statement and Guidelines, published on 11 December 2019, contains guidance for members of AMRAB and expert advisory panels to identify, disclose and manage relevant interests. The policy aims to make members aware that they must disclose interests, current and historic (within the last five years), and promote public confidence in the management of the MRFF. The policy states Health's intention to make a register of interest available to the public. This has not yet occurred.29
2.69 The ANAO reviewed AMRAB meeting minutes and found that most meetings considered conflicts of interest as a standing agenda item. No material personal interests were declared or additional management strategies required for matters under consideration.30
2.70 Health maintains registers of interests declared by AMRAB and expert advisory panel members. Some members have declared many possible or perceived interests while others have recorded none. There is also inconsistency in how often the registers are updated. The ANAO suggests that Health updates the registers for any changes in declarations and makes them available to the public to increase transparency of members' declared interests and their management.
2.71 NHMRC and BGH advised that grant assessment committee members are required to declare any conflicts of interest and, where a conflict of interest is declared in regard to any application, that member does not receive, and is not involved in consideration of, the application. NHMRC and BGH report to Health annually that:
- grant selection committees are applying an appropriate conflict of interest policy; and
- there is compliance with the CGRGs and other requirements.
3. Grants for medical research and medical innovation
Areas examined
This chapter examines whether the Department of Health’s (Health) management of grants under the Medical Research Future Fund (MRFF) is consistent with the requirements of relevant legislation and policy, including the Medical Research Future Fund Act 2015 (MRFF Act).
Conclusion
Health’s management of grants of financial assistance to support medical research and medical innovation is largely consistent with the MRFF Act and the Commonwealth Grant Rules and Guidelines 2017 (CGRGs). There is no evidence of how the design of the MRFF 10-year Plan was influenced by the Australian Medical Research and Innovation Strategy (MRFF Strategy) and Australian Medical Research and Innovation Priorities (MRFF Priorities). Health has suitable arrangements in place to provide assurance that grant applications are assessed on their merit and represent value for money.
Areas for improvement
The ANAO has made one recommendation aimed at ensuring that the reporting of grant opportunities corresponds with the way in which they are published.
3.1 Through MRFF grants, the Australian Government provides financial support for medical research and medical innovation. When managing MRFF grants, including selecting grant recipients, Health and its implementation partners must comply with the MRFF Act, as well as other legislative and policy requirements relating to the provision of grants.
3.2 To assess whether Health’s management of grants under the MRFF Act is consistent with relevant legislation and policy, the ANAO examined:
- whether the MRFF Strategy and the MRFF Priorities guided the development of the 10-year Plan;
- the consistency of grant opportunities with legislative and policy requirements; and
- the processes that Health has in place to provide assurance that approved grants are consistent with the grant opportunity guidelines and represent value for money.
Did the Medical Research Future Fund Strategy and Priorities guide the development of the initiatives in the 10-year Plan?
There is no direct relationship between the initiatives in the 10-year Plan and the MRFF Strategy and MRFF Priorities and it is not clear how the 10-year Plan was designed.
3.3 Under section 32A of the MRFF Act, the Minister for Health (Health Minister) must take the MRFF Priorities into account in making decisions about the financial assistance that is provided from the MRFF Special Account.31 The MRFF Priorities must be consistent with the MRFF Strategy and are intended to help guide the government’s decisions on future research initiatives. The Priorities state that:
While Government must take the MRFF Priorities into consideration when making these decisions, they are not required to address each and every priority.32
3.4 The MRFF Act requires that the MRFF’s Priorities are updated every two years. A comparison of the Priorities for 2016–2018, 2018–2020 and 2020–2022 is shown at Appendix 5. The 19 priorities in 2016–2018 were reduced to 12 in the Priority Statements for 2018–2020 and 2020–2022.33 This reflected feedback AMRAB received from its public consultation on the 2016–2018 Priorities.
3.5 The 10-year Plan represents the government’s $5 billion MRFF investment plan for to 2027–28. It was announced by the government in the 2019–20 Budget and incorporates MRFF initiatives previously approved and initiatives under the National Health and Medical Industry Growth Plan, which the government announced in the Budget and under which plan it had committed $1.3 billion from MRFF over ten years. It also builds on initiatives the government had previously implemented to response to the 2016–2018 MRFF Priorities.34 Some initiatives in the early years of the program represented grants to specific organisations.
3.6 The ANAO reviewed the advice provided to the government on the 10-year Plan. In proposing the new initiatives to be included in the plan (that is, initiatives that were in addition to previously announced initiatives), Health indicated the relevant MRFF Strategy and MRFF Priorities the initiatives would support, but provided no explanation of how the 10-year Plan had been developed. Health has not provided evidence of the basis for its design. There is no direct relationship between the MRFF strategic platforms or MRFF Priorities and the 10-year Plan initiatives.
3.7 The Health Minister reported to Parliament on the 2016–2018 and 2018–2020 Priorities in September 2019 and June 2021 respectively. The reports included a ‘map’ of the initiatives and grant opportunities to the Priorities that were in force during those periods. No grants were funded through one of the initiatives (Research Data Infrastructure) in either period, and some MRFF Priorities are generic and can be attributed to grant opportunities under different initiatives.
Is Health’s management of grant opportunities consistent with legislative and policy requirements?
Health’s management of grant opportunities has been largely consistent with legislative and policy requirements, including the MRFF Act and the CGRGs. However, Health has not consistently advised the Health Minister of the MRFF Priorities that the proposed grant opportunities would address and its approach to reporting the outcomes of grant opportunities reduces transparency. Health provides little information on priorities being considered for future grant opportunities.
3.8 The process to approve and manage MRFF grant opportunities is summarised at Figure 3.1. In summary, the Health Minister approves all grant opportunities on the recommendation of Health and the Health delegate approves all grants.35
Figure 3.1: Health’s process to approve and manage MRFF grant opportunities
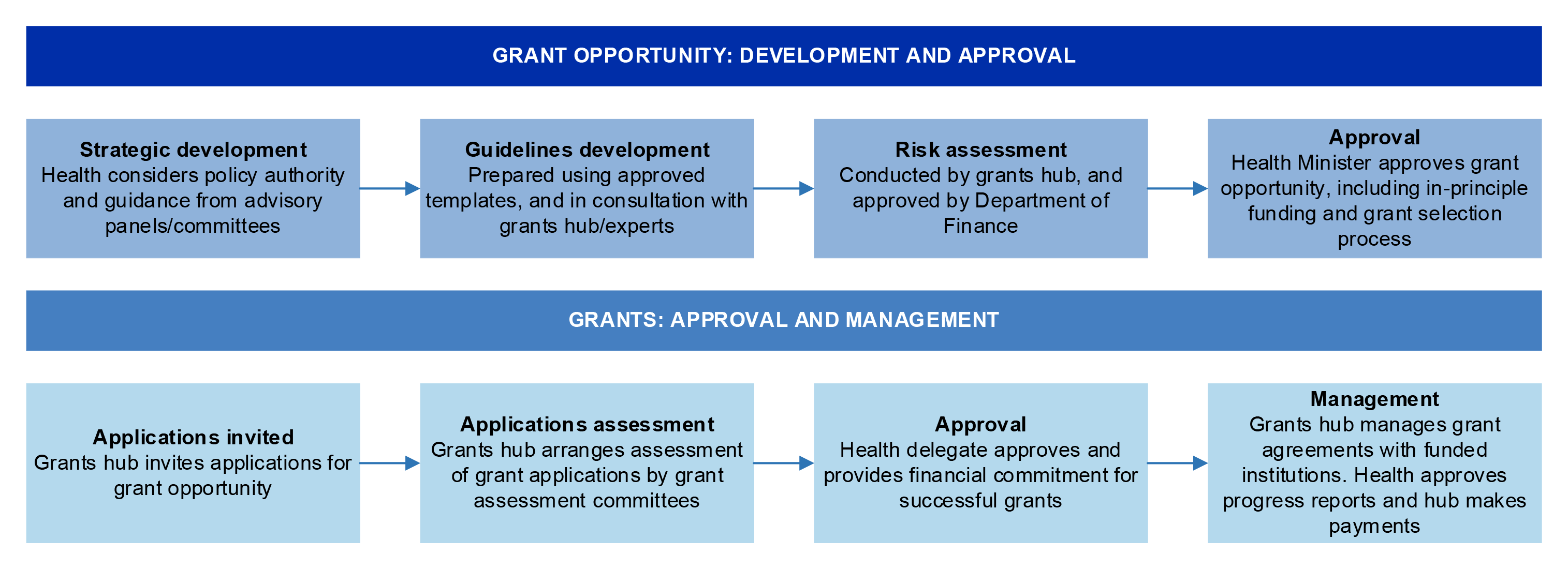
Source: ANAO analysis.
3.9 All MRFF grant opportunities must comply with the MRFF Act and the CGRGs apply (an exception is grants channelled to the state and territories through the Council of Australian Governments (COAG) Reform Fund, none of which have occurred to date). There is an overarching requirement under section 15 of the Public Governance, Performance and Accountability Act 2013 (PGPA Act) for an accountable authority to govern the affairs of the entity in a way that promotes the proper use and management of public resources for which the authority is responsible. This section examines:
- how MRFF grant opportunities are selected;
- the processes that Health has in place to comply with the MRFF Act and the CGRGs; and
- the accessibility of MRFF grant opportunities to potential applicants.
3.10 In reviewing Health’s compliance with the legislative and policy requirements, the ANAO examined a sample of 69 grants (from 46 grant opportunities) that had been approved from the start of the program in 2015 to 2 December 2020.
Advice on grant opportunities
3.11 Grant opportunities are approved by the Health Minister on advice from Health. Health advised the ANAO that a number of factors influence the objectives and scope of grant opportunities. These include:
- advice from relevant experts within Health, such as the Pharmaceutical Benefits Advisory Committee and the Medical Services Advisory Committee;
- for the Research missions in the 10-year Plan, the advice of eight expert advisory panels and committees. Six of the eight expert advisory panels have developed proposed roadmaps, which identify priority areas for investment and implementation plans for each mission and have sought feedback on them, including from international experts; and
- the outcomes of Senate inquiries.
3.12 For non-mission initiatives, Health identifies potential areas for investment, with a focus on gaps in existing investments, research gaps (such as diagnostics and aetiology), areas of unmet need (such as rural health needs and low survival rates) and consideration of potential capacity within the sector. Health may provide options for the Health Minister on grant opportunity priorities.
3.13 In the sample of grants examined by the ANAO there was evidence that the above factors were considered.
3.14 Health has not published an explanation of how grant opportunities are identified or a list of priorities for future research and innovation, particularly for non-mission initiatives. Nineteen of the 33 submissions received by the ANAO considered that it is unclear how grant opportunities are selected and seven submissions said that this gave rise to perceptions of bias.
3.15 Paragraph 32E(3)(a) of the MRFF Act requires AMRAB to take into account ‘the burden of disease on the Australian community’ in determining the MRFF Priorities. Sixteen of the 33 submissions received by the ANAO considered that diseases with a high disability burden, such as asthma, musculoskeletal diseases and neurological and psychiatric disorders, had been overlooked or had received limited coverage.36 Some submissions suggested that primary care merited greater priority in funding allocations, given the prominence of primary care research in the 2020–2022 MRFF Priorities and that it is the largest discipline in the health sector. Aboriginal and Torres Strait Islander healthcare was another area that was identified in submissions as meriting a higher priority and there was a concern from other contributors that funding should be available for basic research.
Compliance with the Medical Research Future Fund Act
Taking account of MRFF Priorities
3.16 As noted at paragraph 3.3, under section 32A of the MRFF Act, the Health Minister or the delegate must take the MRFF Priorities into account in making decisions about financial assistance. Subsection 15A(2) of the MRFF Act states that, in determining whether to require the Minister for Finance (Finance Minister) to debit an amount from the MRFF Special Account to the MRFF Health Special Account, the Health Minister must, among other things, take into account the MRFF Priorities that are in force. Consistent with this, one of 16 MRFF funding principles (Principle 2) is that disbursements from MRFF will reference the MRFF Strategy and related Priorities.
3.17 The ANAO’s examination of the sample of 69 approved grants found that until mid-2019, when seeking the Health Minister’s approval to a grant opportunity, in half of the cases Health advised the Minister of the strategic platforms in the MRFF Strategy and relevant MRFF Priorities that had been taken into account in proposing the grant opportunity.37
3.18 Between mid-2019 and early 2021 the ANAO found from the grants examined that Health’s advice to the Health Minister only indicated the relevant policy approval (generally the 10-year Plan) for the opportunity and did not indicate the strategic platforms in the MRFF Strategy and relevant MRFF Priorities that had been taken into account in proposing the grant opportunity.38 In early 2021 arrangements were revised to ensure that this advice is provided to the Minister.
3.19 The release of funds from the MRFF Special Account to the MRFF Health Special Account is normally requested on a quarterly basis to meet expected payments in the coming quarter for grants that have already been approved or are expected to be approved. In requests to the Finance Minister or to the Department of Finance (Finance) for the release of funds from the MRFF Special Account to the MRFF Health Special Account, to meet the section 15A MRFF Act requirement, the Health Minister or the relevant Health delegate stated that they had taken into account the MRFF Priorities in force at the time. The letters to the Finance Minister and to Finance, and the supporting minutes to the Health delegate relating to these requests, did not include an explanation of what consideration was given to the MRFF Priorities.
Grants to eligible institutions
3.20 Under section 17 of the MRFF Act, the MRFF Special Account can be debited for:
- channelling grants to the COAG Reform Fund to make grants of financial assistance to states and territories;
- channelling grants to the MRFF Health Special Account to make grants of financial assistance to certain bodies; and
- making grants of financial assistance directly to corporate Commonwealth entities.
3.21 As at 31 December 2020 no grants had been channelled to the COAG Reform Fund and no MRFF payments had been made to corporate Commonwealth entities. All grants had been made from the MRFF Health Special Account.
3.22 On 17 December 2021 the government announced that it was contributing, through an appropriation in the 2020–21 Additional Estimates, supplementary funding of $172.5 million for MRFF in 2021–22.39 This MRFF supplementation was provided because the low Reserve Bank of Australia cash rate had reduced the amount available to MRFF to $455 million in 2021–22, compared with an estimated funds availability of $627.5 million.40 Health has advised that grants will be funded direct from this appropriation because the funds are not held in the MRFF Health Special Account.
3.23 Under section 24 of the MRFF Act, grants for the purposes of supporting medical research and medical innovation can be made to a:
- medical research institute;
- university;
- corporate Commonwealth entity; and
- corporation.
3.24 Health has awarded three grants to two local health districts (state government entities established as a body corporate by Part 1 of Chapter 3 of the NSW Health Services Act 1997). One of the grants to the first local health district was a direct grant for a fellowship, which concludes in December 2021, and for which grant applications were originally sought through the National Health and Medical Research Council’s (NHMRC) grants program. The other grant to that organisation was made via the University of Sydney to reduce the risk of Health entering into an agreement with an organisation that could later be deemed ineligible to receive the grant as a ‘corporation’.41 The grant to the second local health district was made in 2017. Health has opted not to enter into any further agreements with local health districts.
3.25 Health has also awarded grants to two state government departments under the 2019 Rural, Regional and Remote Clinical Trial Enabling Infrastructure grant opportunity. The contract negotiations for these grants had not been finalised as at 30 June 2021. The guidelines for this grant opportunity listed a state and territory government or a state or territory government entity as eligible institutions. This was based on Health’s expectation that it could provide funding to a successful state or territory entity under section 20 of the MRFF Act. However, section 20 does not apply to payments to a state or territory through competitive grants processes. As a result, since late 2020 Health has restricted all grant opportunities to institutions that are eligible under section 24 of the MRFF Act. To enable grants to be made to the two state government entities, Health is proposing to make the payments from the MRFF supplementary appropriation (that is, outside the MRFF Act).
Grants to organisations supporting research projects
3.26 Two of the MRFF funding principles are:
8. Encourage partnerships in merit-based collaborative research to engage lateral and fresh thinkers and ideas, and enhance skill and knowledge combinations; and
9. Consider favourably proposals that have collaboration, translation and scalability features to ensure the MRFF is transformative and effort is enduring.
3.27 Consistent with these principles, Health has provided grants to three organisations to initiate and administer research projects in specified medical research fields.42
Compliance with CGRG requirements
3.28 The ANAO assessed Health’s compliance with a number of key requirements of the CGRGs and Finance guidance. T he assessment, shown at Table 3.1, found that Health was generally compliant with the CGRGs.
Table 3.1: Compliance with the CGRGs
|
Requirement |
Detail |
Assessment |
|
Grant opportunities CGRG paragraph 4.4 |
Officials must:
|
◆ Guidelines were developed for all new grant opportunities. These were agreed with Finance for competitive grant opportunities. Commitment approvals note that the activities are a grant for the purposes of the CGRGs and are in accordance with Finance guidance. |
|
Commitment of money by accountable authority or official CGRG paragraph 4.5 |
Where an accountable authority or an official approves the proposed commitment of relevant money in relation to a grant, the accountable authority or official who approves it must record, in writing, the basis for the approval relative to the grant opportunity guidelines and the key principle of achieving value with relevant money. |
◆ This was done in all sampled commitment approvals. |
|
Advice to Ministers CGRG paragraph 4.6 |
Where Ministers exercise the role of an approver, officials’ advice to the Minister must:
|
◆ Health approved grants in all but one instance in the sample. In this instance, the Health Minister approved the grants. Health met the requirement for the advice it provided to the Health Minister and the Minister approved the grant recommended by the selection committee. |
|
Third parties CGRG paragraph 4.8 |
Where a third party administers grants on behalf of the Commonwealth, the relevant accountable authority must ensure the arrangement is in writing and promotes the proper use and management of other Consolidated Revenue Fund money. In addition, the accountable authority must ensure the arrangement requires the third party to apply the CGRGs. |
◆ Health administers all MRFF grants, including through its implementation partners. |
|
Requirements for Ministers CGRG paragraph 4.10 |
Where the proposed expenditure relates to a grant or group of grants, the Minister:
|
◆ The Health Minister approved grant opportunities. Except in one instance, the Health Minister has not approved grants. |
|
Public reporting CGRG paragraphs 5.2, 5.3, 5.5, 5.8 |
Grant opportunity guidelines must be made publicly available on GrantConnect.b From 31 December 2017 an entity must report, on GrantConnect, information on individual grants no later than 21 calendar days after the grant agreement for the grant takes effect. Officials must identify whether a grant agreement contains confidentiality provisions. Information on individual grants must be retained on an entity’s website for at least two financial years. |
▲ Grant opportunity guidelines for all competitive grant opportunities in 2020–21 were made available on GrantConnect.b In 2020–21, 83 per cent of the grants were published within the required 21 days of the grant agreement taking effect. Information on individual grants since the inception of the program is available on Health’s website. |
Key: ◆ Met
▲ Partially met
Note a: The seven CGRG key principles for grants administration are: robust planning and design; collaboration and partnership; proportionality; an outcomes orientation; achieving value with relevant money; governance and accountability; and probity and transparency.
Note b: Guidelines for one-off or ad hoc grants are not required to be published, although Finance encourages entities to publish them on GrantConnect for transparency purposes and Health typically does this.
Source: CGRGs and ANAO analysis of Health documents.
Accessibility of grant opportunities
Competitive and non-competitive grant opportunities
3.29 Principles 4 and 7 of the MRFF funding principles are:
4. Ensure funding is provided utilising a structured contestable process to ensure the highest quality ideas, talent and projects are identified; and
7. Appreciate the role of competitiveness in the research sector as a means of identifying great potential and innovation, and raising Australia’s international research reputation.
3.30 These are consistent with the CGRGs. Paragraph 11.5 of the CGRGs states, in part:
Competitive, merit-based selection processes can achieve better outcomes and value with relevant money. Competitive, merit-based selection processes should be used to allocate grants, unless specifically agreed otherwise by a Minister, accountable authority or delegate. Where a method, other than a competitive merit-based selection process is planned to be used, officials should document why a different approach will be used.
3.31 The way in which Health reports grants on its website by grant opportunity type is shown at
Table 3.2: Health’s categorisation of grants by grant opportunity type
|
Category |
Description |
|
Competitive |
Funding allocated through an openly advertised MRFF opportunity and through which applicants are independently assessed against published assessment criteria. |
|
Competitive pre-qualified |
Funding allocated to a specific organisation following its selection in an open competitive application process outside MRFF. Examples include Advanced Health Research and Translation Centres (assessed by NHMRC), ARC Special Research Initiative, or Industry Growth Centre (assessed by the Department of Industry, Innovation and Science). |
|
Targeted |
Funding is awarded to a specific applicant following assessment of its application against predefined criteria. Grants are sub-categorised as: clinical leadership; national expansion/co-contribution; or patient-led. |
Source: Health documents.
3.32 Table 3.3 shows how Health reported, on its website, the 46 grant opportunities in the ANAO’s sample.
Table 3.3: Sampled grant opportunities reported by Health
|
Grant type |
NHMRC |
BGH |
Cancer Australia |
Total |
|
Competitive |
27 |
7 |
– |
34 |
|
Competitive pre-qualified |
2 |
3 |
– |
5 |
|
Targeted: national expansion/co-contribution |
1 |
1 |
– |
2 |
|
Targeted: patient led |
– |
4 |
– |
4 |
|
Targeted: clinical leadership |
– |
– |
1 |
1 |
|
Total |
30 |
15 |
1 |
46 |
Source: Health grants data for sample, published on its website.
3.33 The reported categorisation is not the same as the way grant opportunities are categorised in the grant opportunity guidelines and as advertised by NHMRC and BGH. The categorisations in the guidelines for the 46 sampled grant opportunities are summarised at Table 3.4.
Table 3.4: Sampled grant opportunities as advertised
|
Grant type |
NHMRC |
BGH |
Cancer Australia |
Total |
|
Open competitivea |
– |
6 |
– |
6 |
|
Targeted competitive or restricted competitiveb |
25 |
1 |
– |
26 |
|
One-off ad hoc / one-off, targeted non-competitive, closed non-competitivec |
3 |
8 |
1 |
12 |
|
N/A - part of NHMRC Fellowships |
2 |
– |
– |
2 |
|
Total |
30 |
15 |
1 |
46 |
Note a: Under section 24 of the MRFF Act, grants for the purposes of supporting medical research and medical innovation can be made to a: medical research institute; university; corporate Commonwealth entity; and corporation. Grant opportunities that were advertised by BGH as ’open competitive’ were restricted to these eligible organisations.
Note b: All ‘competitive’ NHMRC grants ‘are advertised as targeted competitive’ or ‘restricted competitive’ because they are offered to institutions with which NHMRC has an established relationship.
Note c: Grants that were advertised as ‘one-off ad hoc’, ‘one-off’, ‘targeted non-competitive’ and ‘closed, non-competitive’ are all targeted grants.
Source: ANAO analysis of grant opportunity guidelines in sample.
3.34 Using the categorisation of grants in the grant opportunity guidelines (Table 3.4), six of the 46 grant opportunities in the sample were described as open competitive, while 26 were described as targeted or restricted competitive. On its website Health categorised 39 of the grants as competitive.
3.35 In addition, of the five grant opportunities Health classified as ‘competitive pre-qualified’ in the sample examined, four were offered to a single organisation. They were more correctly categorised as ‘one-off ad hoc’ or ‘one-off’ in their grant opportunity guidelines.
3.36 Using different categories for grant opportunities has implications for transparency, as reported outcomes cannot be readily compared to published opportunities. Reporting grant opportunities in the same way in which they are advertised and described in the grant opportunity guidelines and GrantConnect would be more accurate and transparent.
Recommendation no.2
3.37 Department of Health reports grants in the same way that grant opportunities are classified in the grant opportunity guidelines and reported on GrantConnect.
Department of Health response: Agreed.
3.38 The Department is reviewing public reporting of MRFF grants to align with how grant opportunities are classified in the grant opportunity guidelines and reported on GrantConnect.
3.39 In Senate Estimates hearings before the Community Affairs Legislation Committee, Health advised the Committee that around 90 per cent of all grants had been awarded following competitive grant opportunities. While this is the case, as at 30 June 2021 only 64 per cent of all grant opportunities (as distinct from grants) since the inception of the program have been competitive. Competitive and targeted grant opportunities are shown at Figure 3.2.
Figure 3.2: Competitive and targeted grant opportunities by 10-year Plan theme, as at 30 June 2021a
Note a: Competitive and targeted grants are as defined by Health (see Table 3.2).
Note: There were 91 competitive grant opportunities and a total of 142 grant opportunities. However, as part of the Government’s Coronavirus Research Response, grants under the 2020 Antiviral Development for COVID-19 grant opportunity were funded through two different initiatives — the Frontier Health and Medical Research initiative and the Dementia, Ageing and Aged Care initiative.
Source: Department of Health.
3.40 Targeted grants are recommended, for example, when it is Health’s assessment that the organisation(s) to be approached for the grant opportunity are the only one(s) with the necessary skills and expertise to undertake the work or when research funding is required to support an urgent public need, such as COVID-19. This recognises that, in some areas, the availability of the necessary skills and expertise is limited. Projects were also included in the investment plan for an initiative where it was known that a targeted grant opportunity would be required.
3.41 Health is aware, and has advised the Health Minister, that targeted grant opportunities may give rise to criticism by the research sector for a lack of contestability, and that the process lacks transparency and favours some organisations. Even where Health considers that there are a limited number of organisations with the necessary skills and expertise to undertake research in a nominated area, holding competitive merit-based grants rounds may help to avoid bias or perceptions of bias and improve transparency.
3.42 As at 30 June 2021, of the 78 institutions that had received funding through MRFF, 30 (39 per cent) were universities, 27 (35 per cent) were corporations and 16 (21 per cent) were medical research institutes (see Figure 3.3). Thirteen (17 per cent) of the 78 institutions had each received 10 or more grants (receiving in total 72 per cent of all grants) and five (seven per cent) had each received over 40 grants.
Figure 3.3: Institutions to which grants have been awarded, as at 30 June 2021
Source: ANAO analysis of Health grants data.
3.43 Unlike competitive grant opportunities managed by BGH, which allow all organisational types set out in the MRFF Act to apply, competitive grant opportunities through NHMRC are currently only available to NHMRC administering institutions (institutions that meet defined research governance arrangements). This reflects NHMRC’s practice of using overarching agreements with funded institutions to manage agreements under its own funding programs, as well as MRFF. While there are advantages to NHMRC and the funded institutions in this approach, it means that eligible institutions, as defined in the MRFF Act, that are not recognised as administering institutions by NHMRC are currently not able to apply for MRFF grants managed by NHMRC. Health has advised that it is working with NHMRC to broaden the eligibility criteria for grant opportunities that are managed by NHMRC to allow all organisations eligible under section 24 of the MRFF Act to apply for MRFF funding by late 2021.
Timeframes for submitting grant applications
3.44 To provide potential grant applicants with adequate opportunities to submit grant applications, reasonable timeframes for submission of applications need to be provided. In the context of research grants, this recognises that it can take some time to complete the application and often involves collaboration with research partners. For the 34 grant opportunities in the ANAO’s sample that Health described as competitive, the average time for responding was 51 days.43
3.45 Nineteen of the 33 submissions received by the ANAO considered that inadequate timeframes were allowed for the submission of grant applications, with four noting that too many calls for grant applications were made at the end of a year. Four submissions received by the ANAO said that, with the December/January break, this made it difficult to prepare grant applications. There was an expressed preference for grants to be spread more evenly throughout the year and for longer times to be provided to submit applications.
3.46 Five submissions received by the ANAO, one of which was based on a survey of researchers, suggested that Health publish a calendar of proposed grant opportunities with anticipated dates for opportunity openings and deadlines. In mid-2021, Health published a calendar of MRFF grant opportunities on its website, although this calendar does not include information on grant opportunities that are under development.
Does Health have assurance that approved grants are consistent with the grant opportunity guidelines and represent value for money?
Health’s processes for selecting and approving grants provide adequate assurance that grants are assessed consistently with the grant opportunity guidelines and represent value for money.
3.47 Health’s processes for selecting and approving grant applications that are suitable and represent value for money, which are shared with the grants hubs, include:
- determining and publishing selection criteria for each grant opportunity, as part of the grant opportunity guidelines;
- appointing independent and well qualified committees to assess grant applications against the selection criteria;
- providing guidance to grant assessment committees on how grant applications are to be assessed (for example, the rating scale to be used); and
- observing selection committee consideration of grant applications to satisfy itself that the committees’ consideration of proposals has been acceptable.
3.48 Grant assessment committees are appointed to assess the suitability of grant applications for each grant opportunity and to provide scores against each selection criterion. Health has set rating scales, which the grant assessment committees use when assessing the suitability of grant applications.
3.49 Health has set eight core criteria for selecting members of a grant assessment committee.44 The aim is to ensure that, collectively, committees have the expertise to assess grant applications, including industry and commercialisation expertise where appropriate, that they represent the diversity of the Australian population and that they will provide an independent assessment of the grant applications. Health proposes potential grant assessment committee members to NHMRC, but NHMRC is responsible for determining the final composition and appointment of members to grant assessment committees. BGH appoints grant assessment committees based on advice from Health.
3.50 Health has an observer on the grant assessment committees. NHMRC and BGH provide secretariat support for the committees and so also observe the committees’ consideration of the grant applications.
3.51 Health advised the ANAO that its processes are intended to complement, rather than duplicate, NHMRC and BGH processes. For this reason, it has not sought to duplicate NHMRC and BGH privacy, confidentiality or conflicts of interest processes. To provide assurance that applications are independently assessed, assessment committee members are asked to declare any possible conflicts of interest with any grant applications. As noted at paragraph 2.73, where there is a declared conflict of interest, NHMRC and BGH advised the ANAO that the assessment committee member is not provided with copies of the relevant grant applications and has no involvement in the consideration of the application (including absenting themselves from discussion of the applications). This does not necessarily preclude a member of a committee from also being an applicant for a grant. While not explicitly stated in the criteria for panel membership, NHMRC advised that it endeavours to minimise real or perceived conflicts of interest by excluding all applicants to the grant opportunity from participating on the assessment committee, where possible. For MRFF Missions, NHMRC also excludes members of the expert advisory panel from participating on the assessment committee. BGH advised the ANAO that to date Health has specifically declined to allow Chief Investigators45 from participating as a committee member.
3.52 When a committee has finalised its assessment of the applications, NHMRC and BGH provide Health with a list of all applications that meet the funding criteria. Using this assessment and having regard to the available budget, the Health delegate provides the financial commitment for, and approval of, grants for funding. After the delegate has approved the grants, Health advises the Health Minister of the outcome of the selection process for the grant opportunity, who then announces the successful grant applications.
3.53 In the sample of grants examined by the ANAO there was only one instance where Health asked the Health Minister to approve the grants (Health has advised that this is the only instance where the Minister has approved grants). In that instance, the recommended and approved list of grant recipients was prepared by an independent peer review panel of experts. In all other instances, the grants were approved by the Health delegate on the basis of outcomes from grant assessment committees.
3.54 After approval and announcement of the successful grant recipients, unsuccessful applicants are also advised of the outcomes of the grant opportunity. NHMRC advised the ANAO that written feedback is provided for applications that undergo the complete assessment process and BGH advised that they provide feedback orally at the request of the applicant. Eight submissions received by the ANAO expressed dissatisfaction with the level of feedback provided to unsuccessful applicants and a desire for constructive feedback that would assist with possible future grant applications. Health has advised that it has been working with NHMRC and BGH to standardise the feedback that is provided to successful and non-successful applicants.
3.55 Overall the arrangements in place provide Health with adequate assurance that the committees are independently and adequately assessing the grant applications and that approved grant applications are suitable and represent value for money.
4. Performance management
Areas examined
This chapter examines whether the Department of Health (Health) has effectively measured and reported on the performance of funding under the Medical Research Future Fund (MRFF) and evaluated the effectiveness of MRFF financial assistance to support medical research and medical innovation.
Conclusion
Health does not have adequate performance measures for MRFF and has not effectively measured and reported on the performance of MRFF financial assistance in its annual performance statements. Health published a monitoring and evaluation strategy in November 2020, with most of the activities yet to occur. It has also made a number of improvements to the operation of the program.
Area for improvement
The ANAO made one recommendation to improve Health’s reporting of MRFF’s performance.
The ANAO has also suggested that Heath report performance against the 10-year Plan on a financial year basis.
4.1 Commonwealth entities such as Health must measure and report on their performance in achieving their purpose. The performance information developed and reported by Commonwealth entities should meet the requirements specified by the Public Governance, Performance and Accountability Act 2013 (PGPA Act), the associated Public Governance, Performance and Accountability Rule 2014 (PGPA Rule) and resource management guidance provided by the Department of Finance (Finance). The Portfolio Budget Statements (PBS), the corporate plan and the annual performance statements (included in the annual report) are core elements of the Commonwealth performance framework. Application of the Commonwealth performance framework should provide a clear alignment of the entity’s purpose to the planned outcomes of the entity and its performance information to enable the Parliament and the public to consider an entity’s progress towards achieving outcomes.
4.2 To form a conclusion on whether Health’s processes for measuring, reporting and evaluating the performance of the MRFF program are effective, the ANAO examined whether Health has:
- effectively measured and appropriately reported on MRFF’s performance to the Parliament; and
- adequately evaluated the effectiveness of MRFF and established suitable processes to inform continuous program improvement.
Has Health’s management of financial assistance under the Medical Research Future Fund been effectively measured and appropriately reported?
The Minister for Health (Health Minister) provided reports to the Parliament on MRFF financial assistance awarded while the 2016–2018 and 2018–2020 Medical Research and Innovation Priorities (MRFF Priorities) were in force. However, Health does not have adequate performance measures for MRFF against which to report to Parliament on the performance of the program in its annual performance statements and the statements include little analysis of the program’s performance.
4.3 To adequately demonstrate performance, under Section 16EA of the PGPA Rule, Health is required to develop performance measures that:
(a) relate directly to one or more of those purposes or key activities;
(b) use sources of information and methodologies that are reliable and verifiable;
(c) provide an unbiased basis for the measurement and assessment of the entity’s performance;
(d) where reasonably practicable, comprise a mix of qualitative and quantitative measures;
(e) include measures of the entity’s outputs, efficiency and effectiveness if those things are appropriate measures of the entity’s performance; and
(f) provide a basis for an assessment of the entity’s performance over time.
4.4 Finance’s Resource Management Guide No.131, Developing good performance information (RMG 131), provides guidance to entities on the criteria and assessment characteristics of good performance information. Based on this guidance, the ANAO assesses whether measures are relevant, reliable and complete/adequate.46
4.5 Subsection 39 (1) of the PGPA Act requires Health as a Commonwealth entity to prepare and include annual performance statements in its annual report. This performance statement must include an assurance that the performance information provided is an accurate reflection of Health’s performance over the reporting period, and an analysis of the factors that contributed to, or restricted, the delivery of its purpose within the reporting period. The Health Minister also has reporting obligations under the MRFF Act, which requires Health’s support.
Measuring performance
4.6 Health’s PBS for 2019–20, 2020–21 and 2021–22 included measures and targets for MRFF, as shown in Table 4.1. The targets were mirrored in Health’s corporate plans for these years and in its annual report for 2019–20.
Table 4.1: Health’s performance measures and targets for MRFF, 2019–20, 2020–21 and 2021–22
|
2019–20 |
2020–21 |
2021–22 |
|
Performance measures |
||
|
A sustainable source of funding is provided for transformative health and medical research that improves lives, contributes to health system sustainability and drives innovation. |
Provide a sustainable source of funding for transformative health and medical research that improves lives, contributes to health system sustainability and drives innovation. |
Fund transformative health and medical research that improves lives, contributes to health system sustainability, and drives innovation. |
|
Targets |
||
|
Investments announced, grant opportunities offered and grant agreements executed under various MRFF initiatives consistent with the MRFF Act. |
Develop and release the Australian Medical Research and Innovation Priorities 2020–22 and the Australian Medical Research and Innovation Strategy 2021–25. Announce investments, offer grant opportunities and execute grant agreements under various MRFF initiatives consistent with the MRFF Act and the MRFF 10-year Investment Plan. |
Deliver the 2021–22 components of the MRFF 10 Year Investment Plan by announcing investments, offering grant opportunities and executing grant agreements, consistent with the MRFF Act. |
Source: Health’s PBS for 2019–20, 2020-21 and 2021–22.
4.7 In each year, the performance measure is described as funding or providing ‘transformative health and medical research that improves lives, contributes to health system sustainability, and drives innovation’. This is a description of the aim of the MRFF and is not an adequate performance measure. As such, it does not meet the requirements for performance measures.
4.8 Measures should provide information that would enable an assessment to be made of the program’s performance for each financial year. An example would be Health’s performance in delivering on the initiatives in the MRFF 10-year Plan, as reflected to an extent in the target which has been set for 2021–22. The ANAO has recommended that, to effectively measure MRFF’s performance and meet the requirements of the PGPA Rule, Health should develop adequate performance measures for the program (see Recommendation No.3).
Reporting performance
Annual performance reporting
4.9 The PGPA Rule sets out the publication and content requirements to which entities must adhere when preparing annual performance statements and Finance’s Resource Management Guide No.134, Annual performance statements for Commonwealth entities (RMG 134), was developed to assist entities to prepare the statements.
4.10 There are three core minimum requirements that Health must include in its annual performance statements:
- statements (of preparation), which must specify the performance reporting period and an assurance that the performance information provided is an accurate reflection of Health’s performance over the reporting period;
- results achieved, which must demonstrate Health’s performance against its purpose in the relevant reporting period and aligns with the methods of measuring and assessing set out in its corporate plan; and
- analysis of performance, which is analysis of the factors that contributed to, or restricted, the delivery of its purpose within the reporting period.47
4.11 The ANAO examined Health’s annual performance statements for 2019–20 and found that, while the performance criteria and target description for the MRFF are consistent across the corporate plan, PBS and annual performance statements, there was little analysis of the factors that contributed to, or restricted, the delivery of MRFF within the reporting period and there was no prescribed performance methodology. For instance, there was insufficient quantitative and qualitative information about MRFF activities to enable an adequate assessment to be made of the outcomes and impact achieved.
4.12 Health has not reported progress in delivering the 10-year Plan. Reporting actual financial assistance against the 10-year Plan on a financial year basis would provide transparency of progress against the plan.
Recommendation no.3
4.13 Department of Health develops adequate performance measures for the Medical Research Future Fund for inclusion in its portfolio budget statements and annual performance statements.
Department of Health response: Agreed.
4.14 The Department is developing a methodology to enable more effective MRFF reporting to Parliament in the portfolio budget statements and annual performance statements on the performance of MRFF financial assistance, with the intent to enable an assessment of the program’s performance for each financial year and consideration of outcomes and impact achieved.
MRFF Act reporting requirements
4.15 Every two years, following the end of each set of MRFF Priorities, the Health Minister is required under section 57A of the MRFF Act to report to the Parliament on matters relating to the financial assistance provided from the MRFF. The report must include: a description of how the financial assistance provided was consistent with the MRFF Priorities; the processes for determining the grants of financial assistance; and information about any other financial assistance provided by the Commonwealth for medical research and innovation.
4.16 The first round of MRFF Priorities (2016–2018) ceased in November 2018. The Health Minister reported to the Parliament on the financial assistance to support the MRFF Priorities in September 2019, ten months after the first round of MRFF Priorities ceased. The second set of MRFF Priorities (2018–2020) ceased in November 2020. The Minister’s report was tabled in the Parliament in June 2021, six months after the second round of MRFF Priorities ceased. The reports include background on the program, a map of the MRFF initiatives to the MRFF Strategy and MRFF Priorities and details about the grants awarded while the MRFF Priorities were in force. The report on the 2018–2020 MRFF Priorities also includes details of the relevant MRFF priorities addressed by each grant opportunity. However, the reports do not include an analysis of MRFF’s impact on the medical research sector and medical innovation.
Has Health effectively evaluated the effectiveness of the Medical Research Future Fund and established suitable processes in informing continuous progress improvement?
Health published a monitoring, evaluation and learning strategy for MRFF in November 2020, with most of the planned activities in the strategy yet to occur. Health has established suitable continuous improvement processes and has made a number of improvements to the operation of the program.
4.17 Implementing a suitable evaluation framework early with systematic approaches to high-quality performance measurement and evaluation of programs is recognised better practice and informs continuous program improvement. Health uses a range of processes to evaluate performance and inform continuous improvements to the MRFF program, including:
- implementation of an evaluation strategy;
- internal audits; and
- consultation with Health’s implementation partners and with key stakeholders.
Health’s monitoring, evaluation and learning strategy
4.18 Health introduced the MRFF Monitoring, evaluation and learning strategy 2020–21 to in November 2020.48 The strategy, which was developed with the support of AMRAB, outlines the principles for monitoring and evaluating MRFF. It identifies five impact measures and eight measures of success, as shown in Table 4.2.
Table 4.2: MRFF impact and success measures
|
Impact measures |
Measures of success |
|
|
Source: Monitoring, evaluation and learning strategy 2020–21 to 2023–24, November 2020, pp. 7-8.
4.19 The eight success measures are high-level and do not indicate, for example through baselines and targets, what level of improvement is expected and the strategy does not detail what data will be collected to measure performance against the success measures. However, it does indicate that five-yearly impact evaluations will draw on outcomes reported by grant recipients from research projects and evaluations of grant opportunities.
4.20 The strategy proposes the following monitoring, evaluation and learning activities:
- impact evaluations against the eight measures of success, every five years from 2024;
- evaluations of grant opportunities ‘when required to support ongoing program delivery’
- rolling progress reviews or evaluations of research missions (every three years on a rolling basis from 2020–21) and non-mission initiatives (annually from 2019–20);
- annual thematic reports that assess the impact on patients, researchers, missions and translation;
- annual and end-of-activity monitoring and assessment of progress of grant projects; and
- process reviews of financial management and grant administration (initially annually from 2020–21).
4.21 Health did not develop a plan to operationalise the activities outlined in the strategy, but has commenced some activities. In November 2020 Health completed an evaluation of the Rapid Applied Research Translation (RART) initiative and a review of the medical research commercialisation landscape. Health used the recommendations from the RART evaluation to develop the 2020 RART grant opportunity. The 2020 review of the medical research commercialisation landscape presented a summary of stakeholder views on the medical research commercialisation landscape in Australia to help inform future strategies. Health used the findings of this review to develop the guidelines for the 2020 Early Stage Translation and Commercialisation Support grant opportunity. Health is planning to undertake evaluations of the Clinical Trials Activity initiative, the Million Minds Mental Health Research Mission and the Australian Brain Cancer Mission in.
4.22 Most of the medical research and innovation projects funded by MRFF grants have not yet been completed. Nonetheless, development of the monitoring, evaluation and learning strategy and a plan to implement the strategy at the commencement of the program, and not five years after it started, would have enabled some of the planned monitoring and evaluation activities to have commenced earlier and lessons used to inform improvements to the program.
Internal audit
4.23 In 2019 Health undertook an internal audit of the implementation and management of the MRFF program. The audit found that, while the establishment and operation of MRFF were generally compliant with legislative requirements, aspects of MRFF program planning, implementation and administration could be improved. The findings and recommendations of the audit are summarised in Table 4.3. Health closed all of the audit recommendations after receiving details of the action that had been taken to implement them. Analysis in this audit does not support the closure of some of the recommendations.
Table 4.3: Internal audit recommendations
|
Finding |
Recommendation |
|
1. Gaps in the implementation and coordination of MRFF. |
|
|
|
|
|
|
2. Limited performance monitoring and gaps in evaluation planning. |
|
|
|
|
3. Weaknesses in risk management of partnership arrangements. |
|
|
|
|
|
Source: Department of Health, Internal Audit of: Implementation of the Medical Research Future Fund — Final Report, 22 May 2019.
Consultation with Health’s implementation partners and key stakeholders
4.24 Each of the implementation partners informed the ANAO that their roles have evolved over the first five years of the program, and that their respective roles and responsibilities are now clearer. The regular consultation that Health has with NHMRC, BGH and Cancer Australia have also enabled changes to be made to processes, such as revised grant opportunity guidelines and progress reporting templates. Further consideration of the effectiveness of consultation with Health’s implementation partners is at paragraphs 2.22 to 2.33.
4.25 Chapter 2 (paragraphs 2.37 to 2.48) also discussed Health’s consultation with other stakeholders, noting that Health consulted stakeholders on the design of grant opportunities and conflict of interest policy, and consulted mid-career researchers on the drivers affecting their decisions to remain in medical research. In 2020 Health surveyed six key organisations and received four responses on their members’ experiences in applying for, assessing and receiving MRFF competitive grants via NHMRC and BGH as a means of identifying where grant application processes could be improved. Health has considered and has addressed or is addressing almost all comments received.
Appendices
Appendix 1 Entity responses




Appendix 2 Improvements observed by the ANAO
1. The existence of independent external audit exists, and the accompanying potential for scrutiny, improves performance. Improvements in administrative and management practices usually occur: in anticipation of ANAO audit activity; during an audit engagement; as interim findings are made; and/or after the audit has been completed and formal findings are communicated.
2. The Joint Committee of Public Accounts and Audit (JCPAA) has encouraged the ANAO to consider ways in which the ANAO could capture and describe some of these impacts. The ANAO’s 2021–22 Corporate Plan states that the ANAO’ s annual performance statements will provide a narrative that will consider, amongst other matters, analysis of key improvements made by entities during a performance audit process based on information included in tabled performance audit reports.
3. Performance audits involve close engagement between the ANAO and the audited entity as well as other stakeholders involved in the program or activity being audited. Throughout the audit engagement, the ANAO outlines to the entity the preliminary audit findings, conclusions and potential audit recommendations. This ensures that final recommendations are appropriately targeted and encourages entities to take early remedial action on any identified matters during the course of an audit. Remedial actions entities may take during the audit include:
- strengthening governance arrangements;
- initiating reviews or investigations; and
- introducing or revising policies, strategies, guidelines or administrative processes.
4. In this context, the below actions were observed by the ANAO during the course of the audit. It is not clear whether these actions and/or the timing of these actions were planned in response to proposed or actual audit activity. The ANAO has not sought to obtain assurance over the source of these actions or whether they have been appropriately implemented.
|
Paragraph |
Change to Health’s management of MRFF |
|
2.62 to 2.63 |
Memorandum of understanding between Health and NHMRC updated to address shared risks. Shared risks discussed at Health’s quarterly strategic meetings with NHMRC. |
|
2.66 |
Declaration of interests policy and register implemented for HMRO staff. |
|
3.18 |
To provide evidence on how grant opportunities have taken account of MRFF Priorities, as required by the MRFF Act, Health’s advice to the Minister for Health on recommended grant opportunities now includes details of the relevant MRFF strategic platforms and MRFF Priorities that will be addressed. |
|
3.43 |
Health has advised that it is working with NHMRC to allow all organisations that are eligible for MRFF grants under section 24 of the MRFF Act to apply. |
Appendix 3 Number and value of grants by theme and initiative in the10-year Plan, as at 30 June 2021
|
Themes and initiatives |
Funding |
Number of grants |
|
Patients |
$564,846,991 |
220 |
|
Clinical Trials Activity |
$212,290,465 |
120 |
|
Emerging Priorities and Consumer Driven Research |
$336,291,526 |
91 |
|
Global Health |
$16,265,000 |
9 |
|
Researchers |
$238,615,326 |
135 |
|
Clinician Researchers |
$55,506,621 |
96 |
|
Frontier Health and Medical Research |
$151,108,705 |
38 |
|
Researcher Exchange and Development within Industry |
$32,000,000 |
1 |
|
Research missions |
$426,595,385 |
179 |
|
Australian Brain Cancer Mission |
$21,791,073 |
8 |
|
Cardiovascular Health Mission |
$57,923,287 |
30 |
|
Dementia, Ageing and Aged Care Mission |
$40,112,654 |
27 |
|
Genomics Health Futures Mission |
$150,532,779 |
43 |
|
Indigenous Health Research Fund |
$56,802,840 |
17 |
|
Million Minds Mental Health Research Mission |
$64,809,460 |
18 |
|
Stem Cell Therapies Mission |
$25,623,912 |
27 |
|
Traumatic Brain Injury Mission |
$8,999,380 |
9 |
|
Research translation |
$549,780,009 |
136 |
|
Medical Research Commercialisation |
$146,300,000 |
7 |
|
National Critical Research Infrastructure |
$152,143,267 |
12 |
|
Preventive and Public Health Research |
$159,722,568 |
72 |
|
Primary Health Care Research |
$13,851,630 |
11 |
|
Rapid Applied Research Translation |
$64,879,998 |
27 |
|
Research Data Infrastructure |
$12,882,546 |
7 |
|
Total |
$1,779,837,711 |
670 |
Source: Department of Health.
Appendix 4 Actual and budgeted amounts against the 10-year Plan, 2018–19 to 2020–21
Actual and budgeted amounts against the 10-year Plan, 2018–19 to 2020–21
|
Initiatives |
2018–19 Budget ($m) |
2018–19 Actual ($m) |
Difference ($m) |
2019–20 Budget ($m) |
2019–20 Actual ($m) |
Difference ($m) |
2020–21 Budget ($m) |
2020–21 Actual ($m) |
Difference ($m) |
|
Patients |
97.825 |
112.541 |
-14.720 |
124.400 |
125.463 |
-1.070 |
140.329 |
150.860 |
-10.531 |
|
Emerging Priorities and Consumer Driven Research |
53.685 |
63.400 |
-9.720 |
54.300 |
53.955 |
0.340 |
71.165 |
84.532 |
-13.367 |
|
Clinical Trials Activity |
42.340 |
47.361 |
-5.020 |
67.100 |
68.146 |
-1.050 |
66.500 |
63.665 |
2.835 |
|
Global Health |
1.800 |
1.780 |
0.020 |
3.000 |
3.362 |
-0.360 |
2.664 |
2.664 |
0.000 |
|
Researchers |
4.324 |
4.919 |
-0.600 |
49.820 |
47.881 |
1.940 |
92.550 |
84.005 |
8.545 |
|
Frontier Health and Medical Research |
– |
– |
– |
20.000 |
20.113 |
-0.110 |
60.000 |
53.645 |
6.355 |
|
Researcher Exchange and Development within Industry |
– |
– |
– |
8.000 |
8.000 |
– |
10.000 |
10.000 |
0.000 |
|
Clinician Researchers |
4.324 |
4.919 |
-0.600 |
21.820 |
19.768 |
2.050 |
22.550 |
20.360 |
2.190 |
|
Research Missions |
47.786 |
44.732 |
3.050 |
122.560 |
120.675 |
1.880 |
193.690 |
185.610 |
8.080 |
|
Australian Brain Cancer Mission |
8.998 |
4.749 |
4.250 |
5.000 |
7.425 |
-2.430 |
5.000 |
5.000 |
0.000 |
|
Million Minds Mental Health Research Mission |
5.000 |
6.195 |
-1.200 |
5.000 |
6.972 |
-1.970 |
15.000 |
11.955 |
3.045 |
|
Genomics Health Futures Mission |
8.788 |
8.788 |
– |
38.560 |
37.529 |
1.030 |
86.690 |
87.440 |
-0.750 |
|
Dementia, Ageing and Aged Care Mission |
10.000 |
10.000 |
– |
17.500 |
13.122 |
4.380 |
17.500 |
16.991 |
0.509 |
|
Indigenous Health Research Fund |
15.000 |
15.000 |
– |
22.500 |
22.500 |
– |
22.500 |
18.803 |
3.697 |
|
Stem Cell Therapies Mission |
– |
– |
– |
6.000 |
6.000 |
– |
18.000 |
17.624 |
0.376 |
|
Cardiovascular Health Mission |
– |
– |
– |
23.000 |
22.127 |
0.870 |
24.000 |
23.798 |
0.202 |
|
Traumatic Brain Injury Mission |
– |
– |
– |
5.000 |
5.000 |
– |
5.000 |
3.999 |
1.001 |
|
Research Translation |
54.200 |
44.174 |
10.030 |
77.800 |
81.505 |
-3.710 |
171.200 |
177.460 |
-6.260 |
|
Preventive and Public Health Research |
18.800 |
11.153 |
7.650 |
32.500 |
35.187 |
-2.690 |
57.100 |
63.860 |
-6.760 |
|
Primary Health Care Research |
– |
– |
– |
5.000 |
6.706 |
-1.710 |
5.000 |
7.146 |
-2.146 |
|
Rapid Applied Research Translation |
20.000 |
17.621 |
2.380 |
20.000 |
16.630 |
3.370 |
20.000 |
20.630 |
-0.630 |
|
Medical Research Commercialisation |
15.400 |
15.400 |
– |
15.300 |
15.300 |
- |
35.300 |
60.300 |
-25.000 |
|
National Critical Research Infrastructure |
– |
– |
– |
5.000 |
7.682 |
-2.680 |
43.800 |
12.642 |
31.158 |
|
Research Data Infrastructure |
– |
– |
– |
– |
– |
– |
10.000 |
12.883 |
-2.883 |
|
2018–19 Budget Balance over the forward estimates |
18.248 |
|
|
0.123 |
|
|
0.166 |
|
|
|
Revised profile |
|
|
|
18.000 |
|
|
|
|
|
|
Total |
222.383 |
206.366 |
16.017 |
392.703 |
375.524 |
17.179 |
597.935 |
597.935 |
0.000 |
Note: The indicated budget for 2019–20 reflects $18.0 million that was later shifted via a movement of funds into the 2020–21 financial year.
Source: Department of Health.
Appendix 5 Comparison of the Medical Research Future Fund Strategy and Priorities, 2016 to 2022
|
2016-21 MRFF Strategy strategic platforms |
2016–18 Priorities |
2018–20 and 2018–22 Priorities |
|
Strategic and international horizons |
Antimicrobial resistance International collaborative research Disruptive technology
|
One Health —Antimicrobial Resistance Global health and health security Aboriginal and Torres Strait Islander health Ageing and aged care |
|
Data and infrastructure |
Clinical quality registries National data management study MRFF infrastructure and evaluation Communicable disease control |
Digital health intelligence
|
|
Health services and systems |
National Institute of Research Building evidence in primary care Behavioural economics application Drug effectiveness and repurposing |
Comparative effectiveness research Primary care research
|
|
Capacity and collaboration |
National infrastructure sharing scheme Clinical researcher fellowships Industry exchange fellowships |
Consumer-driven research Clinical researcher capacity
|
|
Trials and translation |
Clinical trial network Public good demonstration trials Targeted translation topics |
Drug repurposing Public health interventions
|
|
Commercialisation |
Research incubator hubs Biomedical translation |
Translational research infrastructure
|
Source: ANAO analysis of MRFF Priorities for 2016–2018, 2018–2020 and 2020–2022.
Footnotes
1 Medical Research Future Fund Act 2015, section 3.
2 In addition to the MRFF, the Future Fund Board of Guardians manages five other public asset funds. Available from https://www.futurefund.gov.au/ [accessed 1 December 2021]
3 Department of Finance, Medical Research Future Fund, available from https://www.finance.gov.au/government/australian-government-investment-funds/medical-research-future-fund [accessed 1 December 2021].
4 While the MRFF Act also provides that funds can be transferred to the COAG Reform Fund and directly to corporate Commonwealth entities for MRFF grants, no such grants have been made.
5 Preamble to and section 3 of the Medical Research Future Fund Act 2015.
6 Medical Research Future Fund Act 2015, section 3.
7 In addition to MRFF, the Future Fund Board of Guardians manages five other public asset funds. Available from https://www.futurefund.gov.au/ [accessed 20 July 2021]
8 Department of Finance, Medical Research Future Fund, available from https://www.finance.gov.au/government/australian-government-investment-funds/medical-research-future-fund [accessed 1 December 2021]
9 Because of its responsibility for managing the MRFF Special Account, the Finance Minister is responsible for the MRFF Act under the Administrative Arrangements Order of 18 March 2021.
10 While the MRFF Act also provides that funds can be transferred to the COAG Reform Fund and directly to corporate Commonwealth entities for MRFF grants, no such grants have been made.
11 The Investment Funds Legislation Amendment Bill 2021 (introduced into Parliament on 25 August 2021) proposes that the duration of the MRFF Strategies change from five years to six years and the duration of the MRFF Priorities change from two years to three years. These changes will enable every second iteration of the MRFF Priorities to coincide with the commencement of a new MRFF Strategy.
12 Previous Priority Statements were for 2016–2018 and 2018–2020.
13 Cancer Australia initially managed some grants, but has not done so since 2018.
14 Preamble to and section 3 of the Medical Research Future Fund Act 2015.
15 Subsection 15(1) of the Public Governance, Performance and Accountability Act 2013.
16 The terms of reference for the eight expert advisory panels established by the Health Minister ‘to guide’ the MRFF 10-year Plan research missions are publicly available. The eight panels and committees are: Australian Brain Cancer Mission Expert Strategic Advisory Group; Cardiovascular Health Mission Expert Advisory Panel; Dementia, Ageing and Health Care Mission Expert Advisory Panel; Genomics Health Futures Mission Expert Advisory Committee; Indigenous Health Research Fund Expert Advisory Panel; Million Minds Mental Health Research Mission Expert Advisory Panel; Stem Cell Therapies Mission Expert Advisory Panel; and the Traumatic Brain Injury Mission Expert Working Group.
17 Department of Health, Australian Medical Research Advisory Board (AMRAB), available from https://www.health.gov.au/committees-and-groups/australian-medical-research-advisory-board-amrab [accessed 4 June 2021].
18 Department of Health, Budget 2019–20: Investing in Health and Medical Research – Health and Medical Research Office, available from https://www.health.gov.au/resources/publications/budget-2019-20-investing-in-health-and-medical-research-health-and-medical-research-office [accessed 22 July 2021].
19 The Brain Cancer Mission is one of eight Research Missions established under the 10-year Plan. The relevant Health line areas provide similar support for the seven other missions.
20 Medical Research Future Fund — Australian Medical Research and Innovation Strategy 2016–2018, p.3.
21 NHMRC website, available from https://www.nhmrc.gov.au/funding/new-grant-program/overview [accessed 1 June 2021].
22 This practice ceased in early 2020 and all funding is now provided through other MRFF grant opportunities.
23 While there are exceptions, the standard grant requirement is for progress reports to be provided annually and final reports within six months of the completion of a project. Few projects have been completed to date and so few final reports are available.
24 NHMRC, BGH and Cancer Australia provide advice to Health on progress reports submitted by grant recipients. Health approves all progress payments for BGH grants. For NHMRC grants, Health advises NHMRC of any issues that need to be followed up before progress payments are made.
25 A list of all closed consultation activities is on Health’s website under its consultation hub: available from https://consultations.health.gov.au/consultation_finder/?keyword=medical+research+future+fund [accessed 1 September 2021].
26 Formerly the COAG Health Council.
27 Health’s Risk Management Policy identifies eight themes and assigns risk tolerance levels to each.
28 Department of Finance, Commonwealth Risk Management Policy, 2014, p. 16.
29 Auditor-General Report No.5 2019–20 Australian Research Council’s Administration of the National Competitive Grants Program. The ANAO found that the Council has a Research Management System (RMS) to help manage conflicts of interest by recording and tracking individual assessor and participant relationships. The RMS prevents assessors being assigned to a proposal where there is a known relationship with any named participant or organisation on the proposal (p. 8).
30 Section 32K of the MRFF Act requires an AMRAB member to disclose a material personal interest in a matter being considered or about to be considered by AMRAB. The member must as soon as possible after the relevant facts have come to the member’s knowledge disclose the nature of the interest at an AMRAB meeting and to the Health Minister. A disclosure to the AMRAB meeting must be recorded in the minutes of the meeting.
31 MRFF Act, part 2, section 10.
32 Australian Medical Research and Innovation Priorities 2020-2022, p. 1, available from https://www.health.gov.au/resources/publications/australian-medical-research-and-innovation-priorities-2020-2022 [accessed 1 September 2021].
33 The 2018-2020 and 2020-2022 Priorities are almost identical. The Chair of AMRAB advised that no changes were proposed because of the limited opportunity to consult widely during the COVID-19 pandemic and the fact that the MRFF Strategy will be updated in 2021 for the coming five years.
34 The government’s previous MRFF initiatives are listed in: Australian Government, Medical Research Future Fund: Financial Assistance to Support the Australian Medical Research and Innovation Priorities 2016–18, Table 5, pp. 12–13, available from https://www.health.gov.au/resources/publications/financial-assistance-to-support-the-australian-medical-research-and-innovation-priorities-2016-2018 [accessed 1 September 2021].
35 On 17 May 2017 the Health Minister delegated his powers under sections 15A (debiting amounts from the MRFF special account to the MRFF Health special account), 26 (debiting of amounts from the MRFF Health special account for the payment of grants) and 27 (entering into agreements with grantees other than a state or territory) of the MRFF Act to designated Health and NHMRC delegates.
36 These submissions referred to an article by Gilbert et al, which found that, for grants awarded between 2016 and September 2019, there was a strong association between MRFF funding and death burden, with many non-fatal conditions receiving comparatively little funding. Stephen E Gilbert, Rachelle Buchbinder, Ian A Harris and Christopher G Maher, A comparison of the distribution of Medical Research Future Fund grant with disease burden in Australia, Medical Journal of Australia, 2021.
37 In the ANAO’s sample of 69 grants there were 46 grant opportunities, of which 22 were approved before 30 June 2019. For 11 grant opportunities the recommendations to the Health Minister mentioned the specific strategic platforms and MRFF priorities that the opportunities addressed. For six opportunities the advice to the Health Minister noted that the grant opportunity had been guided by the Strategy and Priorities or that the Strategy and Priorities had been considered. For five opportunities no mention was made of the Strategy or Priorities.
38 For the 24 grant opportunities in the sample approved after 30 June 2019, the advice to the Health Minister referenced the specific strategic platforms and priorities in only two and no mention was made of the MRFF Strategic or Priorities for 13 opportunities.
39 G Hunt (Minister for Health and Aged Care), $172.5 million funding top up to support Australian medical research, media release, 17 December 2020, available from https://www.health.gov.au/ [accessed 1 September 2021].
40 Portfolio Additional Estimates Statements 2020–21 — Health Portfolio, ‘Medical Research Future Fund Supplementation’, pp.11 and 25.
41 The Investment Funds Legislation Amendment Bill 2021 (introduced into Parliament on 25 August 2021) proposes a definition of ‘corporation’ to be included in the MRFF Act.
42 These are:
- a grant of $10 million in 2017 under which ten research projects were funded, prioritised on how well they met the objectives of the Boosting Preventive Health Research Program;
- a grant of $5 million in 2017 for the Australian Young Cancer Patient Clinical Trials Initiative for four clinical trials to improve outcomes for young cancer patients; and
- three grants to one organisation with a total value of $154 million.
43 The average is based on the time between the date the grant opportunity was opened and the date it was closed, as outlined in each grant opportunity guidelines. The timeframe does not include earlier ‘soft’ releases of guidelines by BGH. The sample included grant opportunities where a rapid turnaround was required (for COVID-19 and bushfire-related opportunities).
44 These criteria are: trans-disciplinary research; academic researchers; clinical expertise; health services delivery and implementation; research translation; health services and implementation expertise; consumers and patients groups advisor; and Aboriginal and Torres Strait Islander health expertise.
45 Chief investigators are the proposed leaders of research projects in MRFF grant applications.
46 ANAO, Audit Insights — Performance Measurement and Monitoring – Developing Performance Measures and Tracking Progress, November 2020, available from https://www.anao.gov.au/work/audit-insights/performance-measurement-and-monitoring-developing-performance-measures-and-tracking-progress [accessed 1 September 2021].
47 Department of Finance, Resource Management Guide No. 134: Annual performance statements for Commonwealth entities, July 2017, pp. 11–26.
48 Medical Research Future Fund, Monitoring, evaluation and learning strategy 2020–21 to 2023–24, November 2020, available from https://www.health.gov.au/resources/publications/mrff-monitoring-evaluation-and-learning-strategy-2020-21-to-2023-24 [accessed 1 September 2021].